All content on this site is intended for healthcare professionals only. By acknowledging this message and accessing the information on this website you are confirming that you are a healthcare professional. If you are a patient or carer, please visit Know AML.
The AML Hub website uses a third-party service provided by Google that dynamically translates web content. Translations are machine generated, so may not be an exact or complete translation, and the AML Hub cannot guarantee the accuracy of translated content. The AML Hub and its employees will not be liable for any direct, indirect, or consequential damages (even if foreseeable) resulting from use of the Google Translate feature. For further support with Google Translate, visit Google Translate Help.
The AML Hub is an independent medical education platform, sponsored by Daiichi Sankyo, Johnson & Johnson, Syndax, Thermo Fisher Scientific, Kura Oncology, and AbbVie. Funders are allowed no direct influence on our content. The levels of sponsorship listed are reflective of the amount of funding given. View funders.
Now you can support HCPs in making informed decisions for their patients
Your contribution helps us continuously deliver expertly curated content to HCPs worldwide. You will also have the opportunity to make a content suggestion for consideration and receive updates on the impact contributions are making to our content.
Find out more
Create an account and access these new features:
Bookmark content to read later
Select your specific areas of interest
View AML content recommended for you
Menin inhibitors in AML: Where are we now, and where are we going?
Do you know... Which of the following is a key challenge in designing trials of menin inhibitors for the treatment of NPM1m AML to meet regulatory requirements?
During a meeting of the AML Hub Steering Committee, held on February 19, 2026, Jorge Cortes chaired a discussion on the topic, Menin inhibitors in AML: Where are we now, and where are we going? The discussion featured contributions from Charles Craddock, Brian Huntly, and Jeffrey Lancet.
Menin inhibitors in AML: Where are we now, and where are we going?
Menin inhibitors in AML: Where are we now, and where are we going?
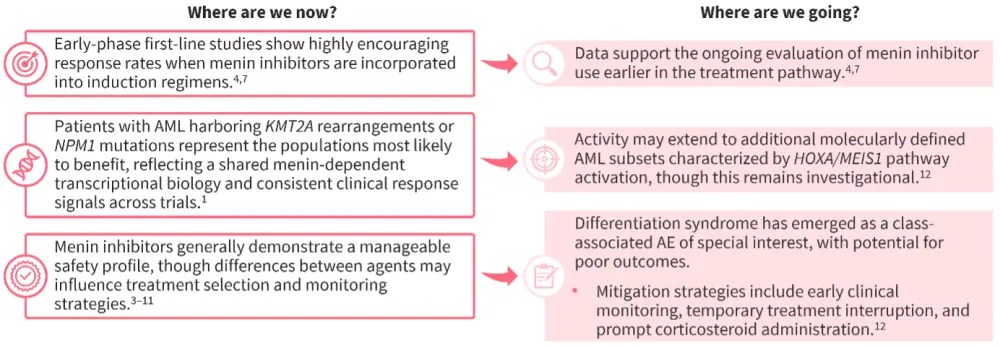
Cortes first outlined the rationale for the use of menin inhibitors in acute myeloid leukemia (AML) and their mechanism of action (Figure 1). He then provided an overview of those in development for the treatment of AML and the latest data from key clinical trials, including updates from the 67th American Society of Hematology (ASH) Annual Meeting and Exposition and the European Hematology Association (EHA) 2025 Congress (Table 1). He concluded by discussing potential future directions for menin inhibition in AML, such as their use earlier in the treatment pathway or in additional molecularly defined AML subsets, and strategies to mitigate differentiation syndrome (Figure 2).
Figure 1. Rationale for menin inhibition in AML, and mechanism of action1,2
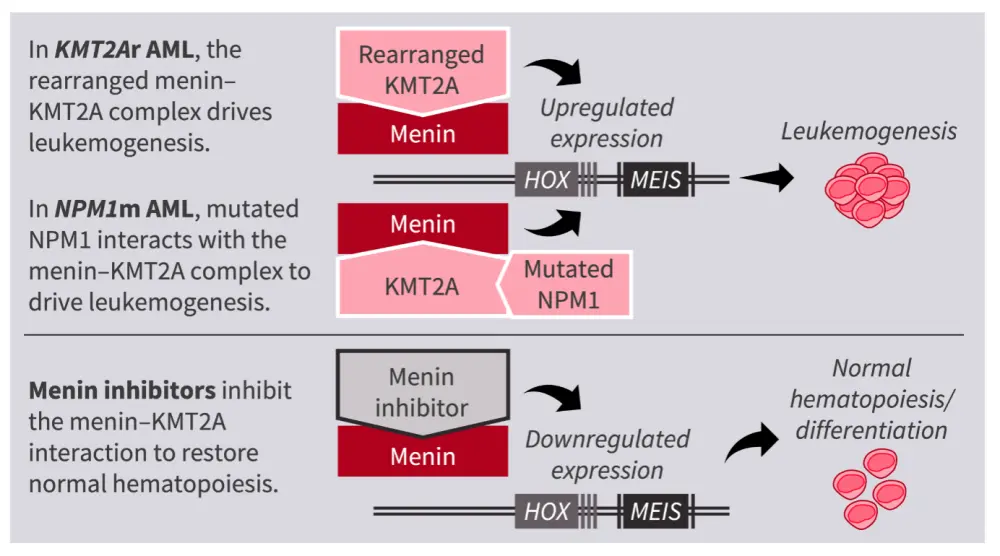
Table 1. Menin inhibitors in AML: An overview of the latest updates from ASH 2025 and EHA 2025
Key trials | Intervention | Population | Efficacy population, n | ORR, % | CR/CRh rate, % | Safety population, n | Grade ≥3 TEAEs, % |
(phase I/II; NCT04065399) | monotherapy | R/R NPM1m or KMT2Ar AML | 78 | 66.7 | 23.1 | 95 | 57.9* |
SAVE4 (phase I/II; NCT05360160) | Revumenib + decitabine/ | ND NPM1m AML | 14 | 86.0 | 79.0 | 21 | -§ |
| ND KMT2Ar AML | 7 | 86.0 | 86.0 | ||||
(phase I/II; NCT04067336) | Ziftomenib monotherapy | R/R NPM1m AML | 92 | 33 | 22 | 92 | 93 |
KOMET-0076,7 (phase I; NCT05735184) | Ziftomenib + Ven + Aza | R/R NPM1m AML | 48 | 65 | 40 | 83 | 40* |
| R/R KMT2Ar AML | 32 | 41 | 22 | ||||
| ND NPM1m AML | 37 | 89 | 78 | 40 | 40* | ||
(phase I/II; NCT04811560) | Bleximenib monotherapy | R/R NPM1m or KMT2Ar AML | 21† | 47.6† | 33.3† | 31† | 22.6*† |
ALE10029,10 (phase I/II; NCT05453903) | Bleximenib + Ven + Aza | R/R NPM1m AML | 12 | 91.7 | 41.7 | 49 | 96 |
| R/R KMT2Ar AML | 10 | 70.0 | 40 | ||||
| ND NPM1m AML | 16 | 93.8 | 62.5 | ||||
| ND KMT2Ar AML | 4 | 75.0 | 75 | ||||
Bleximenib + 7+3 | ND NPM1m AML | 15 | 100 | 73.3 | 25 | 100 | |
| ND KMT2Ar AML | 9 | 88.9 | 88.9 | ||||
DSP-5336-10111 (phase I/II; NCT04988555) | monotherapy | R/R KMT2Ar AML | 15‡ | 73.3‡ | 40‡ | 116 | -§ |
| R/R NPM1m AML | 25 | -§ | -§ |
*Treatment-related adverse events. †Data reported for the RP2D, 90/100 mg BID. ‡Data reported for the RP2D, 300 mg BID. §Values not available.
AML, acute myeloid leukemia; ASH, American Society of Hematology; Aza, azacitidine; BID, twice daily; CR, complete remission; CRh, CR with partial hematologic recovery; EHA, European Hematology Association; KMT2Ar, KMT2A-rearranged; ND, newly diagnosed; NPM1m, NPM1-mutated; ORR, overall response rate; RP2D, recommended phase II dose; R/R, relapsed/refractory; TEAE, treatment-emergent adverse event; Ven, venetoclax.
Figure 2. Potential future directions for menin inhibition for the treatment of AML
Key points
- Potential future directions for menin inhibitors in AML include investigation of existing salvage regimens in the first-line setting, in both fit and unfit patient populations.
- Careful consideration of key challenges in the NPM1-mutated (NPM1m) and KMT2A-rearranged (KMT2Ar) settings will be required when designing trials, to meet regulatory requirements.
- Randomized controlled trials in NPM1m AML are needed for regulatory approval. However, as current standard therapy already gives favorable outcomes in this patient population, there is a need to demonstrate sufficient benefit with any investigational regimens vs existing regimens, with regard to efficacy, safety, associated costs, and treatment burden.
- Designing randomized controlled trials for KMT2Ar AML is difficult due to its rarity. The use of historical controls may be acceptable to regulators if a large and clinically meaningful benefit is demonstrated, given the poor prognosis of this population; however, adjusting for allogeneic hematopoietic stem cell transplantation (allo-HSCT) will be an important issue.
- Resistance to menin inhibitors is an ongoing challenge in treating patients with NPM1m or KMT2Ar AML.
- In cases of genetic resistance, where certain mutations may confer resistance to specific inhibitors, some new agents may be advantageous by inducing degradation of the target protein rather than targeting the gene itself.
- In the more common instances of non-genetic resistance, which often involves reactivation of leukemogenic programs via alternative epigenetic regulators (e.g. PRC1 complex, KAT6A pathways), more inhibitors targeting these pathways exist, thus offering potential combination regimens as a strategy for mitigating the clinical impact of resistance.
- Careful patient selection for trials investigating triplet regimens is another consideration, as early use of novel triplet regimens could promote resistance and compromise future salvage options.
- Risk stratification can facilitate identification of patients most likely to benefit when designing early-phase trials.
This educational activity is independently supported by Johnson & Johnson. All content was developed by the faculty in collaboration with SES. Funders were allowed no influence.
References
Please indicate your level of agreement with the following statements:
The content was clear and easy to understand
The content addressed the learning objectives
The content was relevant to my practice
I will change my clinical practice as a result of this content
Your opinion matters
Which AML-related topic do you currently need the most practical guidance on?


 Jeffrey Lancet
Jeffrey Lancet Charles Craddock
Charles Craddock Brian Huntly
Brian Huntly Jorge Cortes
Jorge Cortes.webp&w=3840&q=75)

.webp&w=3840&q=75)