All content on this site is intended for healthcare professionals only. By acknowledging this message and accessing the information on this website you are confirming that you are a healthcare professional. If you are a patient or carer, please visit Know AML.
The AML Hub website uses a third-party service provided by Google that dynamically translates web content. Translations are machine generated, so may not be an exact or complete translation, and the AML Hub cannot guarantee the accuracy of translated content. The AML Hub and its employees will not be liable for any direct, indirect, or consequential damages (even if foreseeable) resulting from use of the Google Translate feature. For further support with Google Translate, visit Google Translate Help.
The AML Hub is an independent medical education platform, sponsored by Daiichi Sankyo, Johnson & Johnson, Syndax, Thermo Fisher Scientific, Kura Oncology, and AbbVie. Funders are allowed no direct influence on our content. The levels of sponsorship listed are reflective of the amount of funding given. View funders.
Now you can support HCPs in making informed decisions for their patients
Your contribution helps us continuously deliver expertly curated content to HCPs worldwide. You will also have the opportunity to make a content suggestion for consideration and receive updates on the impact contributions are making to our content.
Find out more
Create an account to access:
Bookmark & personalize site content
Receive alerts for new content in your areas of interest
View AML content recommended for you
Addition of nucleoside analogues to 7+3: A case report of high-risk AML
During the 2nd How to Diagnose and Treat Acute Leukaemia conference of the European School of Haematology (ESH), Nigel Russell delivered an oral presentation on the addition of nucleoside analogues to frontline cytarabine plus anthracyclines (7+3) for the treatment of high-risk acute myeloid leukemia (AML).1 This is the last in a series of case reports about the addition of a third agent to frontline 7+3.
Patient details1
- 39-year old male
- Presentation: White blood cell count of 18 × 109/L
- Cytogenetics:
- Normal karyotype
- NMP1 mutation
- FLT3 wildtype
- DNMT3A wildtype
Diagnosis1
- De novo AML with NPM1 mutation
Management plan1
The patient was included in the NCRI AML19 trial (ISRCTN78449203). His management plan is demonstrated in Figure 1.
Figure 1. Treatment management plan1
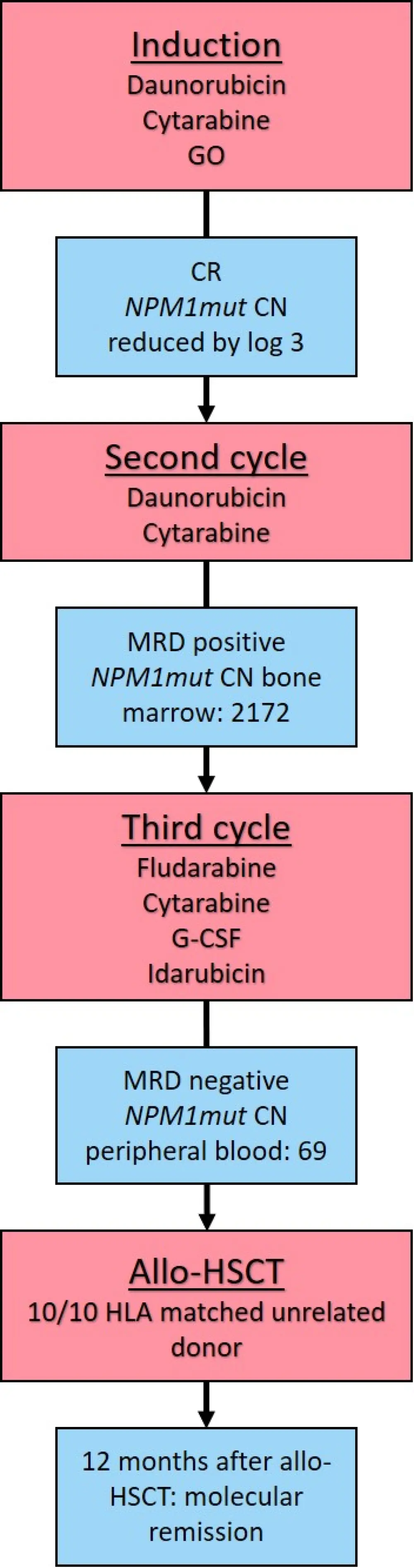
allo-HCST, allogeneic hematopoietic stem cell transplant; CN, copy number; CR, complete response; G-CSF, granulocyte-colony stimulating factor; GO, gemtuzumab ozogamicin; HLA, human leukocyte antigen; MRD, measurable residual disease; NPM1mut, nucleophosmin-1 mutation.
Induction
- Daunorubicin (60 mg/m2 on Days 1, 3, 5)
- Cytarabine (100 mg/m2 twice daily on Days 1–10)
- Gemtuzumab ozogamicin (GO; 3 mg/m2 on Day 1)
|
NPM1 mutations are associated with high CD33 expression. GO is an antibody–drug conjugate, comprising calicheamicin linked to a CD33 monoclonal antibody, that targets CD33+ cells. Therefore, patients with NMP1 mutations are likely to be better responders to GO therapy and this was demonstrated in the combined analysis of the AML16 and 17 trials.2,3 The AML16 trial of GO (3 mg/m2) with/without 7+3, in older adults (> 60 years of age) with treatment-naïve AML, showed that GO significantly improved overall survival (OS) and reduced relapse risk.2 |
|
The AML19 trial (ISRCTN78449203) is an ongoing trial investigating induction with standard chemotherapy (daunorubicin + cytarabine) with the addition of single or fractionated dose GO in patients with AML or high-risk myelodysplastic syndromes (MDS) with favorable, intermediate risk, or unknown cytogenetics, between the ages of 16–60 years (or up to 65 if fit). |
The patient responded to induction therapy with complete remission (CR) at Day 21 (assessed by bone marrow aspiration) and showed good count recovery. He was considered as standard risk on the AML17 risk score.
The NMP1 mutant copy number assessed by qPCR showed a 3-log reduction following induction.
The patient received a second cycle of:
- Daunorubicin
- Cytarabine
After Cycle 2, the patient had positive measurable residual disease (MRD) with a 1-log increase in NPM1 mutant transcript levels (copy number 2172) in the bone marrow. Assessment of the bone marrow showed ongoing morphological remission.
Peripheral blood also showed good count recovery:
- White blood cell count 4.2
- Neutrophils 2.1 × 109/L
- Platelets 301 × 109/L
The patient was entered into the high-risk arm of the AML19 trial and received FLAG-Ida:
- Fludarabine (30 mg/m2 on Days 2–6)
- Cytarabine (2 g/m2 on Days 2–6)
- Granulocyte-colony stimulating factor (263 µg on Days 1–7)
- Idarubicin (8 mg/m2 Days 4–6)
|
After a median follow-up of approximately 5-years, patients in the AML17 trial with negative, low (< 200/105 in blood and < 1000 in bone marrow), and high levels of MRD had an estimated 2-year OS of 83%, 63%, and 13%, respectively (p < 0.0001).4 |
|
The AML17 trial (previously reported on the AML Hub) also compared the use of daunorubicin + clofarabine (DClo) with that of FLAG-Ida in the second course of treatment for patients who were classified as high risk. When comparing DClo vs FLAG-Ida, the final compete response rate (84% vs 87%; p = 0.5) and the percentage of patients that went to transplant (58% vs 53%; p = 0.4) were similar in both treatment arms. There was also a non-significant trend for more patients to be flow MRD-negative in the FLAG-Ida arm (23% vs 30%; p = 0.08). However, there was a superior relapse-free survival and OS for patients with high-risk AML in the FLAG-Ida arm (5-year OS from first CR posttransplant, 41% vs 64%).1 |
Bone marrow MRD assessment after FLAG-Ida showed a reduction in NMP1 copy number to 69 and MRD negativity in the peripheral blood.
|
The AML17 trial further showed, with qPCR of the peripheral blood, that patients who were MRD positive had an extremely high-risk of relapse (86%) compared with those who were MRD negative (34%), which lead to a significant difference in 5-year OS (24% vs 73%, p < 0.001).1 |
HSCT
The patient then received an allogeneic hematopoietic stem cell transplantation (allo-HSCT).
Posttransplant evolution
The patient remains in molecular remission at 12 months after allo-HSCT.
Conclusion
Allo-HSCT remains the treatment of choice for the long-term survival of patients with high-risk AML, however, early recognition of high-risk disease (through MRD measurements) and optimizing induction therapy pre-HSCT allows more patients to remain in CR post-HSCT. Nucleoside analogue-containing regimens, such as FLAG-Ida, have been shown to allow high-risk patients to continue to HSCT, with a lower risk of post-transplant relapse, as demonstrated by this case report.
The first article in this case report series describes the addition of midostaurin to frontline 7+3. Find out more information here. The second case report in this series describes the addition of GO to frontline 7+3.
References
Please indicate your level of agreement with the following statements:
The content was clear and easy to understand
The content addressed the learning objectives
The content was relevant to my practice
I will change my clinical practice as a result of this content
Your opinion matters
Which AML-related topic do you currently need the most practical guidance on?

