All content on this site is intended for healthcare professionals only. By acknowledging this message and accessing the information on this website you are confirming that you are a healthcare professional. If you are a patient or carer, please visit Know AML.
The AML Hub website uses a third-party service provided by Google that dynamically translates web content. Translations are machine generated, so may not be an exact or complete translation, and the AML Hub cannot guarantee the accuracy of translated content. The AML Hub and its employees will not be liable for any direct, indirect, or consequential damages (even if foreseeable) resulting from use of the Google Translate feature. For further support with Google Translate, visit Google Translate Help.
The AML Hub is an independent medical education platform, sponsored by Daiichi Sankyo, Johnson & Johnson, Syndax, Thermo Fisher Scientific, Kura Oncology, and AbbVie. Funders are allowed no direct influence on our content. The levels of sponsorship listed are reflective of the amount of funding given. View funders.
Now you can support HCPs in making informed decisions for their patients
Your contribution helps us continuously deliver expertly curated content to HCPs worldwide. You will also have the opportunity to make a content suggestion for consideration and receive updates on the impact contributions are making to our content.
Find out more
Create an account to access:
Bookmark & personalize site content
Receive alerts for new content in your areas of interest
View AML content recommended for you
Lenalidomide maintenance in high-risk AML not eligible for an immediate transplant
Despite novel combined approaches and intensive therapy options in treating acute myeloid leukemia (AML), there is still a risk of relapse, particularly in patients with high-risk disease, which may ultimately lead to poor outcomes. Allogeneic stem cell transplantation (allo-SCT) is still considered the ideal option in AML with intermediate risk or adverse cytogenetics, where the risk of relapse is > 35% once remission is reached. However, factors such as age, comorbidities, performance status, and toxicity of previous therapy may prevent patients with AML undergoing allo-SCT. Therefore, there is an ongoing interest and effort to provide an effective option in terms of maintenance therapy for patients who are not candidates for transplant.
Iman Abou Dalle et al. investigated the efficacy and safety of low-intensity, continuous dosing of lenalidomide maintenance in patients with high-risk AML in remission and who were not candidates for immediate allo-SCT in a phase II study (NCT02126553).1 The results were recently published in Cancer, and here, we are pleased to summarize the study design, and key outcomes.
Phase III QUAZAR AML-001 trial (NCT01757535) investigated an oral hypomethylating agent (CC-486) as a maintenance option which led to the U.S Food and Drug Administration (FDA) approval for this setting, for more information click here.
Study design
A single arm, open-label, phase II study.
Eligibility criteria:
- ≥ 18 years of age
- High-risk AML in the first complete remission (CR1) or second complete remission (CR2) following induction chemotherapy and at least one consolidation cycle and not a candidate for immediate allo-SCT
(high-risk AML: adverse cytogenetics [a FLT3 mutation], history of a myeloid neoplasm, secondary AML [sAML], significant dysplasia in the bone marrow, therapy-related AML [tAML], primary refractory AML requiring > 1 cycle of intensive induction chemotherapy to achieve CR1, persistent measurable minimal residual disease [MRD] following the initial induction therapy)
- Eastern Cooperative Oncology Group (ECOG) performance status of ≤ 3
- Adequate bone marrow reserve (absolute neutrophil count > 0.5 × 109/L and platelet count ≥ 30 × 109/L)
- Primary endpoint: Relapse-free survival (RFS) after achieving remission
- Secondary endpoints: Overall survival (OS), the duration of remission, and safety
- The treatment plan is depicted in Figure 1.
Figure 1. Study treatment1
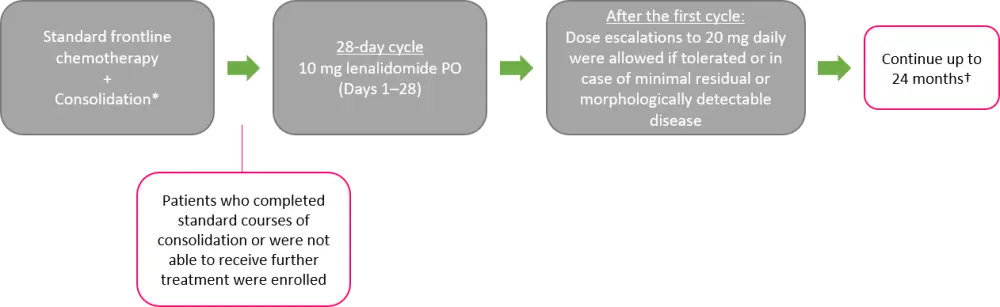
PO, per oral; SCT, stem cell transplantation.
*Adequate number of cycles at the discretion of primary physician.
†Unless in cases of clinically significant treatment failure, progression, or unacceptable toxicity, where proceeding to SCT or discontinuation is considered the best option for the patient.
Patients
- The number of patients enrolled in the study was 28. Table 1 summarizes the baseline characteristics
- At the time of enrollment, 89% of patients were in CR1, and 11% were in CR2
- Median time to lenalidomide maintenance was 8.8 months (4–19.5 months) from the start of induction therapy, and 5.8 months (2.4–12 months) from the first or second remission date
Table 1. Baseline characteristics1
|
AML, acute myeloid leukemia; HMA, hypomethylating agent; MDS, myelodysplastic syndrome; MPN, myeloproliferative neoplasms; MRD, minimal residual disease; SCT, stem cell transplantation; tAML, therapy-related AML. |
|
|
Characteristic |
N = 28 |
|---|---|
|
Median age, years (range) |
61 (24–87) |
|
Male sex, % |
50 |
|
Median number of consolidation cycles (range) |
4 (1–10) |
|
High-risk feature, % |
|
|
MRD persistence |
54 |
|
Adverse cytogenetics |
21 |
|
Prior MDS/MPN |
25 |
|
Prior HMA therapy |
14 |
|
AML with myelodysplastic changes |
14 |
|
Adverse molecular profile |
25 |
|
Second remission |
11 |
|
Primary refractory |
7 |
|
tAML |
7 |
|
Prior SCT |
7 |
|
Molecular profile, % |
|
|
FLT3-ITD/D835 |
7 |
|
IDH1/IDH2 |
25 |
|
DNMT3A |
25 |
|
NPM1 |
21 |
|
NRAS/KRAS |
18 |
|
TET2 |
18 |
|
TP53 |
7 |
|
RUNX1 |
3 |
|
ASXL1 |
3 |
|
Cytogenetics, % |
|
|
Diploid |
57 |
|
Complex (> 2 abnormalities) |
7 |
|
Monosomal |
11 |
|
KMT2A rearranged, t(4;11) |
4 |
|
Miscellaneous |
21 |
Results
Of 28 patients, 10 (36%) completed 24 months of lenalidomide maintenance, and one patient was still on lenalidomide at the time of analysis; none of them relapsed after a median follow-up of 16 months (0–32.7 months). The median follow-up duration was 22.5 months (2.6–55 months) and the median treatment duration was 8 cycles (1–24 cycles).
- The reasons for treatment discontinuation included relapse (n = 12) after a median of 3 months (0.7–23 months) and toxicity (n = 5)
- among those who relapsed, five patients had sAML, and two patients had tAML
- Patients who started lenalidomide within or beyond 6 months of remission had a median duration of remission of 4.5 months and 25.7 months, respectively
- Two-year OS and RFS from enrollment were 63% and 50%, respectively
- patients with sAML or tAML had a median OS and RFS of 6.7 and 2.5 months, respectively; for those with other high-risk features, the median OS and RFS were not reached (p < 0.0001)
- Of five patients who had MRD at the start of lenalidomide maintenance, four relapsed in 3.7 months (1.2–5.7 months), and one underwent allo-SCT and remained in remission.
Safety
All patients were included in the safety analysis.
- The most common (≥15% occurrence) treatment-related adverse events (TRAEs) included rash, pruritus, fatigue, diarrhea, and nausea/vomiting; most were Grade 1–2 in severity.
- Grade 3 TRAEs were as follows:
- Rash: 18%
- Fatigue, thrombocytopenia, and nausea/vomiting: 7% each
- Febrile neutropenia, dyspnea, and stroke: 3% each
- Grade 4 TRAEs included neutropenia (14%) and thrombocytopenia (7%)
- Lenalidomide dose could be escalated to 15 mg in two patients
- Dose reduction to 5 mg was required in 12 patients due to rash, thrombocytopenia, neutropenia, nausea, and diarrhea in descending order of frequency
- Treatment was discontinued in five patients (due recurrent rash, n = 3; Grade 2 dyspnea, n = 1; stroke after one cycle, n = 1) following a median of seven cycles (range, 1–20); none of these patients relapsed within a median follow-up of 10.8 months (6.6–22.4 months)
Conclusion
These findings demonstrated that lenalidomide maintenance was effective with a good tolerability in patients with high-risk AML who were ineligible for transplant. However, investigators concluded that this approach may not be as effective in eliminating continuous MRD, with a median time to relapse of 3.7 months, compared with MRD-negative patients. This may translate into a benefit in managing relapses in MRD-negativity. The median RFS was 23 months and the OS was not reached, which were longer than previous experience in patients with high-risk AML who did not undergo allo-SCT. The median duration of remission (17.3 months) was also longer than previously reported. sAML or tAML were associated with a significantly shorter duration of remission and OS compared with other high-risk features.
Lenalidomide maintenance was limited to 24 months and the authors acknowledged that a longer therapy might be an issue considering the increased risk of secondary hematologic and solid malignancies observed in multiple myeloma.
References
Please indicate your level of agreement with the following statements:
The content was clear and easy to understand
The content addressed the learning objectives
The content was relevant to my practice
I will change my clinical practice as a result of this content
Your opinion matters
Which AML-related topic do you currently need the most practical guidance on?

