All content on this site is intended for healthcare professionals only. By acknowledging this message and accessing the information on this website you are confirming that you are a healthcare professional. If you are a patient or carer, please visit Know AML.
The AML Hub website uses a third-party service provided by Google that dynamically translates web content. Translations are machine generated, so may not be an exact or complete translation, and the AML Hub cannot guarantee the accuracy of translated content. The AML Hub and its employees will not be liable for any direct, indirect, or consequential damages (even if foreseeable) resulting from use of the Google Translate feature. For further support with Google Translate, visit Google Translate Help.
The AML Hub is an independent medical education platform, sponsored by Daiichi Sankyo, Johnson & Johnson, Syndax, Thermo Fisher Scientific, Kura Oncology, and AbbVie. Funders are allowed no direct influence on our content. The levels of sponsorship listed are reflective of the amount of funding given. View funders.
Now you can support HCPs in making informed decisions for their patients
Your contribution helps us continuously deliver expertly curated content to HCPs worldwide. You will also have the opportunity to make a content suggestion for consideration and receive updates on the impact contributions are making to our content.
Find out more
Create an account to access:
Bookmark & personalize site content
Receive alerts for new content in your areas of interest
View AML content recommended for you
How cytogenetic risk impacts the outcome of allogeneic stem cell transplantation
Cytogenetic profile is a predictor of outcome in patients with acute myeloid leukemia (AML). It also helps with the choice of the best post-remission therapeutic approach.
However, limited data are available on the impact of cytogenetic risk stratification on the outcome of allogeneic hematopoietic cell transplantation (allo-HCT). Here, we report the results from two studies evaluating the prognostic value of cytogenetic risk stratification in patients undergoing allo-HCT with active disease and in patients with FLT3-mutated, NPM1-negative (FLT3+ NPM1–) AML undergoing allo-HCT in remission.
Impact of cytogenetic risk in patients undergoing allo-HCT with active disease1
A retrospective study, investigating the impact of cytogenetic risk on the outcomes of allo-HCT in patients with relapsed/refractory (R/R) AML, included patients aged ≥ 18 years who underwent their first allo-HCT in 2000─2017 with active disease, and who had cytogenetics available at time of diagnosis. Poiani, Battipaglia et al. recently published the results in American Journal of Hematology.1
The primary endpoint of the study was overall survival (OS).
Patient characteristics
The baseline characteristics of the patients according to cytogenetic risk group are reported in Table 1.
Table 1. Patient characteristics according to cytogenetic risk group1
|
allo-HCT, allogeneic hematopoietic cell transplantation; auto-, autologous; MAC, myeloablative conditioning; PS, performance status; RIC, reduced intensity conditioning; TCD, T-cell depletion. |
||||
|
Characteristic |
Favorable |
Intermediate |
Adverse |
p value |
|---|---|---|---|---|
|
Median age at allo-HCT, years (range) |
46 (19─75) |
52 (18─77) |
50 (19─78) |
< 0.01 |
|
Median time from diagnosis to allo-HCT, months (range) |
13 (2─132) |
6 (1.5─161) |
5 (1.5─54) |
< 0.01 |
|
Disease status at allo-HCT, % |
|
|
|
< 0.01 |
|
Previous auto-HCT, % |
6 |
6 |
1 |
< 0.01 |
|
Karnofsky PS at allo-HCT, % |
|
|
|
0.58 |
|
Conditioning regimen, % |
|
|
|
0.77 |
|
Type of donor, % |
|
|
|
< 0.01 |
|
In vivo TCD, % |
52 |
51 |
59 |
0.01 |
|
Median follow-up, months (range) |
40 (1─167) |
51 (1–195) |
49 (1─179) |
0.90 |
Results
The main outcomes are shown in Figure 1. The median time to neutrophil engraftment was 15 days for all three cytogenetic groups (p = 0.41) and no difference was observed in the cumulative incidence of engraftment (p = 0.07). In addition, no differences were observed according to cytogenetic risk in non-relapse mortality (NRM) and Grade II─IV acute graft-versus-host disease (aGvHD).
Figure 1. Outcomes according to risk group1
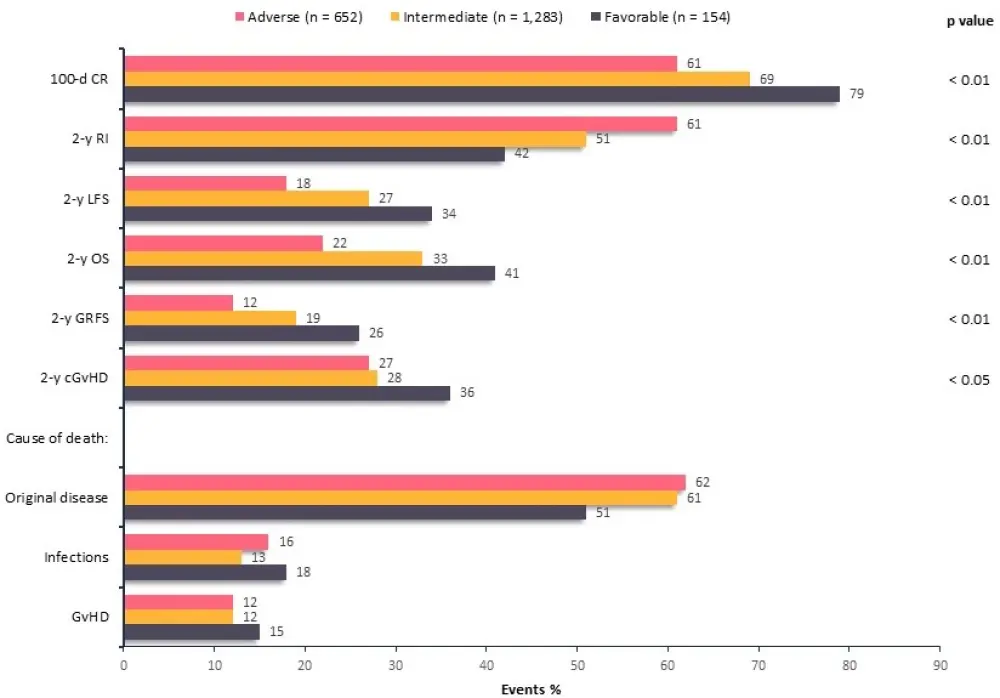
cGvHD, chronic GvHD; CI, confidence interval; d, day; CR, complete remission; GRFS, graft-versus-host disease/relapse-free survival; GvHD, graft-versus-host disease; LFS, leukemia-free survival; OS, overall survival; RI, relapse incidence; y, year.
The multivariate analysis (Table 2) confirmed the univariate results, with patients in the intermediate and adverse groups having worse outcomes compared to patients in the favorable group.
Table 2. Results of multivariate analysis for the main outcomes1
|
aGvHD, acute GvHD; BM, bone marrow; cGvHD, chronic GvHD; CI, confidence interval; CMV, cytomegalovirus; GRFS, GvHD/relapse-free survival; GvHD, graft-vs-host disease, HLA, human leukocyte antigen; HR, hazard ratio; LFS, leukemia-free survival; NRM, non-relapse mortality; OS, overall survival; PBSC, peripheral blood stem cell; PS, performance status; RI, relapse incidence; TCD, T-cell depletion; UD, unrelated donor. |
|||
|
Outcome |
Variables |
HR (95% CI) |
p value |
|---|---|---|---|
|
RI |
Favorable risk (reference) |
|
|
|
Primary refractory (reference) |
|
|
|
|
Patient age (incremental age 10 years) |
0.93 (0.88─0.99) |
0.02 |
|
|
Year of transplant |
0.98 (0.96─1.00) |
< 0.02 |
|
|
Karnofsky PS ≥ 90% |
0.79 (0.67─0.92) |
< 0.01 |
|
|
Center effect |
|
< 0.01 |
|
|
NRM |
Primary refractory (reference) |
|
|
|
Patient age (incremental age 10 years) |
1.18 (1.07─1.29) |
< 0.01 |
|
|
Karnofsky PS ≥ 90% |
0.57 (0.45─0.71) |
< 0.01 |
|
|
LFS |
Favorable risk (reference) |
|
|
|
Primary refractory (reference) |
|
|
|
|
Karnofsky PS ≥ 90% |
0.70 (0.62─0.80) |
< 0.01 |
|
|
Center effect |
|
< 0.02 |
|
|
OS |
Favorable risk (reference) |
|
|
|
Primary refractory (reference) |
|
|
|
|
Karnofsky PS ≥ 90% |
0.65 (0.57─0.75) |
< 0.01 |
|
|
Patient positive CMV serology vs negative |
1.16 (1.00─1.34) |
< 0.05 |
|
|
GRFS |
Favorable risk (reference) |
|
|
|
Primary refractory (reference) |
|
|
|
|
Karnofsky PS ≥ 90% |
0.73 (0.65─0.82) |
< 0.01 |
|
|
In vivo TCD: Yes vs No |
0.84 (0.73─0.97) |
< 0.02 |
|
|
Grade II─IV aGvHD |
Patient age (incremental age 10 years) |
0.92 (0.85─0.99) |
< 0.03 |
|
HLA-identical sibling donor (reference) |
|
|
|
|
UD 10/10 |
1.67 (1.31─2.13) |
< 0.01 |
|
|
UD 9/10 |
1.9 (1.41─2.55) |
< 0.01 |
|
|
Female donor to male recipient vs other |
1.30 (1.03─1.65) |
< 0.03 |
|
|
PBSC vs BM |
1.69 (1.08─2.66) |
< 0.03 |
|
|
In vivo TCD: Yes vs No |
0.64 (0.50─0.82) |
< 0.01 |
|
|
Center effect |
|
< 0.01 |
|
|
cGvHD |
Year of transplant |
0.96 (0.94─0.99) |
< 0.01 |
|
PBSC vs BM |
2.21 (1.29─3.77) |
< 0.01 |
|
|
In vivo TCD: Yes vs No |
0.63 (0.49─0.82) |
< 0.01 |
|
No significant differences in transplant outcomes were observed according to conditioning intensity but, when analyzed separately the outcomes according to the conditioning intensity, patients in the adverse and intermediate groups and those transplanted with active disease due to relapse had worse outcomes.
Impact of FLT3-ITD status on transplant outcomes
An analysis performed only in patients with FLT3-ITD status available (FLT3-ITD+, n = 301; FLT3-ITD–, n = 399) showed worse outcomes in FLT3-ITD+ versus FLT3-ITD– patients:
- Relapse incidence (RI), 59% vs 45%
- Leukemia-free survival (LFS), 21% vs 32%
- OS, 24% vs 41%
- GvHD/relapse-free survival (GRFS), 15% vs 23%
Impact of cytogenetic risk in patients with FLT3+ NPM1– AML undergoing allo-HCT in remission2
In cytogenetically normal patients, FLT3+ NPM1– status predicts poor prognosis and serves as indication for allo-HCT. However, the impact of cytogenetics-based risk stratification in patients with FLT3+ NPM1– AML undergoing allo-HCT is still under investigation. During the 2021 Transplantation & Cellular Therapy Meetings of ASTCT and CIBMTR, Arnon Nagler presented the results from a retrospective study evaluating the impact of the EBMT cytogenetic risk score stratification in patients with FLT3+ NPM1– AML undergoing allo-HCT in remission.2
Patients included in the study were ≥ 18 years with de novo AML, underwent first allo-HCT from matched siblings, 9-10/10 unrelated, or haplo donors, were in first or second complete remission between 2000─2019, and had both cytogenetics and FLT3 and NPM1 mutation status available.
The primary endpoint of the study was LFS.
Patient characteristics
The baseline characteristics of the patients according to cytogenetic risk group are reported in Table 3.
Table 3. Patient characteristics according to cytogenetic risk group2
|
CR, complete remission; CR1, first CR; CR2, second CR; HCT, hematopoietic cell transplantation; IQR, interquartile range; MAC, myeloablative conditioning; MSD, matched sibling donor; RIC, reduced intensity conditioning; UD, unrelated donor. |
||||
|
Characteristic |
Good |
Intermediate |
Adverse |
p value |
|---|---|---|---|---|
|
Median age, years (range) |
39.9 (19─68.6) |
49.6 (18─76.8) |
48.2 (18.1─71.1) |
< 0.0001 |
|
Type of donor |
||||
|
MSD/syngeneic, % |
38.4 |
38.2 |
33.5 |
0.15 |
|
Status at HCT, % |
||||
|
CR1 |
66.1 |
91.1 |
90.3 |
< 0.0001 |
|
Karnofsky score, % |
||||
|
< 90 |
23.2 |
20.2 |
23.3 |
0.48 |
|
Conditioning regimen |
||||
|
MAC, % |
63.4 |
56.4 |
60.4 |
0.23 |
|
Median follow-up, months (IQR) |
29.9 (15.5─61.5) |
38.4 (15.7─75.0) |
28.6 (12.5─58.4) |
0.005 |
Results
No differences were observed according to cytogenetic risk in graft failure and engraftment, or in the incidence of Grade II─IV and Grade III─IV aGvHD, chronic (c)GvHD, and NRM. The incidence of extensive cGvHD was higher in patients in the good vs intermediate vs poor risk groups (27% vs 15.7% vs 20.3; p = 0.041), but this was not confirmed in the multivariate analysis.
Significant differences were observed according to cytogenetic risk in RI, LFS, and OS (Table 4).
Table 4. Outcomes according to cytogenetic risk group2
|
CI, confidence interval; HR, hazard ratio; LFS, leukemia-free survival; OS, overall survival; RI, relapse incidence. |
||||||
|
Cytogenetics |
RI |
LFS |
OS |
|||
|---|---|---|---|---|---|---|
|
HR |
p value |
HR |
p value |
HR |
p value |
|
|
Intermediate vs good |
1.82 |
0.005 |
1.48 |
0.02 |
1.43 |
0.052 |
|
Adverse vs good |
1.83 |
0.013 |
1.65 |
0.009 |
1.74 |
0.008 |
These results were confirmed in the multivariate analysis. In addition, the multivariate analysis found that significant differences in GRFS were observed in the adverse vs good risk group (HR, 1.41; 95% CI, 1.03─1.93; p = 0.032).
The main causes of death in the good vs intermediate vs adverse risk groups were:
- Original disease: 42.4% vs 52.6% vs 51.2%
- GvHD: 21.2% vs 18.9 vs 17.9
- Infections: 21.2% vs 15.4% vs 17.9%
Conclusion
- Results from these studies demonstrated that cytogenetic risk is a valid predictor of transplant outcome in both the R/R setting and in patients with FLT3+ NPM1– AML undergoing allo-HCT, and it should be considered in the setting of allo-HCT for risk stratification
- Adverse risk cytogenetics were associated with the worst outcomes
- Regardless of cytogenetic risk, patients transplanted with active disease due to relapse had worse outcomes than those transplanted with active disease due to refractoriness to chemotherapy
References
Please indicate your level of agreement with the following statements:
The content was clear and easy to understand
The content addressed the learning objectives
The content was relevant to my practice
I will change my clinical practice as a result of this content
Your opinion matters
Which AML-related topic do you currently need the most practical guidance on?

