All content on this site is intended for healthcare professionals only. By acknowledging this message and accessing the information on this website you are confirming that you are a healthcare professional. If you are a patient or carer, please visit Know AML.
The AML Hub website uses a third-party service provided by Google that dynamically translates web content. Translations are machine generated, so may not be an exact or complete translation, and the AML Hub cannot guarantee the accuracy of translated content. The AML Hub and its employees will not be liable for any direct, indirect, or consequential damages (even if foreseeable) resulting from use of the Google Translate feature. For further support with Google Translate, visit Google Translate Help.
The AML Hub is an independent medical education platform, sponsored by Daiichi Sankyo, Johnson & Johnson, Syndax, Thermo Fisher Scientific, Kura Oncology, and AbbVie. Funders are allowed no direct influence on our content. The levels of sponsorship listed are reflective of the amount of funding given. View funders.
Now you can support HCPs in making informed decisions for their patients
Your contribution helps us continuously deliver expertly curated content to HCPs worldwide. You will also have the opportunity to make a content suggestion for consideration and receive updates on the impact contributions are making to our content.
Find out more
Create an account and access these new features:
Bookmark content to read later
Select your specific areas of interest
View AML content recommended for you
Quizartinib, decitabine, and venetoclax combination therapy in patients with AML and prior exposure to FLT3 inhibitors
During the European Hematology Association (EHA) 2022 Congress, Yilmaz, et al.1 reported the updated safety and efficacy data from a phase I/II study (NCT03661307), evaluating the triplet combination of quizartinib, a FLT3 inhibitor, with decitabine and venetoclax in patients with FLT3-ITD-mutated acute myeloid leukemia (AML).
This update included data from 35 patients with relapsed/refractory AML (R/R AML) or high-risk myelodysplastic syndromes (n = 28), and newly diagnosed AML (ND AML; n = 7). During the 63rd American Society of Hematology (ASH) Annual Meeting & Exposition, data from 28 patients (R/R AML, n = 23; ND AML, n = 5) were reported, and have been summarized here. We summarize the key points reported by Yilmaz, et al. below.
Study design
The primary objective was to establish the recommended phase II dose (RP2D) of quizartinib combined with decitabine and venetoclax. The secondary objectives aimed to determine the following: complete remission (CR); CR with incomplete count recovery (CRi); minimal residual disease (MRD); and overall survival (OS). The full study design can be seen in more detail in this previous AML Hub article here. As previously reported, 30 mg once daily was the RP2D.
Results
The baseline characteristics of the 35 patients were similar to the previous report (Table 1).
Table 1. Baseline characteristics*
|
AML, acute myeloid leukemia; ASCT, autologous stem cell transplantation; FLT3i, FLT3 inhibitors; HMA, hypomethylating agents; R/R, relapsed/refractory; Ven, venetoclax. |
||
|
Characteristic |
R/R AML |
Frontline |
|---|---|---|
|
Median age, years (range) |
57 (23–86) |
70 (65–85) |
|
Male sex, % |
32 |
14 |
|
AML diagnosis, % |
||
|
De novo |
75 |
14 |
|
Secondary |
14 |
72 |
|
Therapy-related |
11 |
14 |
|
Prior therapies, median (range) |
3 (1–5) |
— |
|
HMA + Ven, % |
53 |
— |
|
≥1 prior FLT3i, % |
82 |
— |
|
≥2 prior FLT3i, % |
32 |
— |
|
Prior gilteritinib, % |
75 |
— |
|
ASCT, % |
32 |
— |
|
Karyotype, % |
||
|
Diploid |
46 |
14 |
|
Adverse |
25 |
43 |
|
Other |
29 |
43 |
Response rates
Of the 28 patients diagnosed with R/R AML, 82% achieved CR; response rates for the patient subgroups are shown in Figure 1. All patients with ND AML achieved composite CR, with a CR rate of 57%. Within the R/R AML cohort, 25% of patients achieved MRD negativity measured by flow cytometry, and 33% achieved MRD negativity by FLT3-PCR. On the other hand, 66% of patients in the frontline cohort achieved MRD negativity by flow cytometry, and 86% of patients achieved FLT3-PCR MRD negativity.
A total of 12 patients (43%) with R/R AML underwent autologous stem cell transplantation (ASCT). The 60-day mortality rate of this group of patients was 11%.
Figure 1. Composite CR rates for patients with R/R AML (n = 28)*
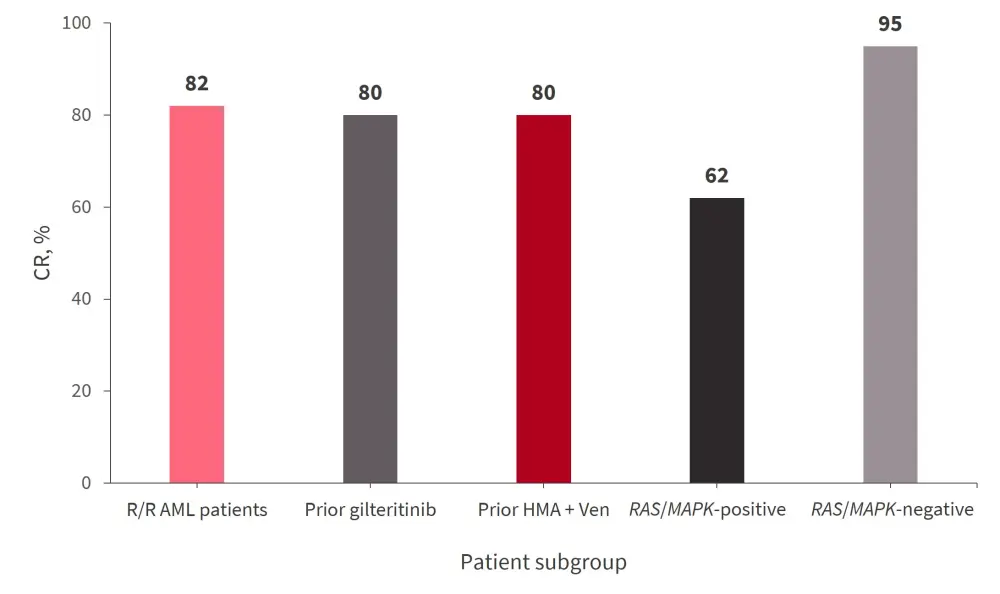
AML, acute myeloid leukemia; CR, complete remission; HMA, hypomethylating agents; R/R, relapsed/refractory; Ven, venetoclax.
*Adapted from Yilmaz, et al.1
In the R/R AML cohort, the median follow-up was 13 months, with a median OS of 6.9 months.
- In patients with prior exposure to gilteritinib, the median OS was 6.3 months, compared to 4.7 months in patients with prior hypomethylating agents + venetoclax inhibitor exposure.
- 12 patients in remission underwent ASCT, of which four were alive at the last follow-up.
- Of the 11 patients in remission who were unable to undergo ASCT, two were alive.
- All five patients with unresponsive disease died due to disease progression.
In the ND AML cohort, the median follow-up was 16 months, and the median OS was 14.5 months.
- Of the three patients in remission who underwent ASCT, one was alive at last follow-up.
- Four patients were in remission but were unable to undergo ASCT; three were alive.
Consistent with the previous report, durable responses (CR >6 months and at last follow-up) were achieved in patients without RAS/MAPK mutations at baseline. On the other hand, patients with RAS/MAPK mutations at baseline did not respond to treatment or relapsed early (within 6 months). The post-therapy next-generation sequencing conducted in 11 patients who achieved a response, and who then relapsed, revealed that four of these patients had emerging RAS/MAPK mutations, two had emerging FLT3-F691L mutations, and three were FLT3-ITD-negative.
Safety
In the phase I part of the study, no patients developed a dose limiting toxicity when treated with the 30 mg once daily dose of quizartinib; however, two patients who received once daily dosing of 40 mg quizartinib developed hematologic dose-limiting toxicities, both of which were Grade 4 myelosuppression. As a result, further enrollment to the 40 mg daily dosing schedule was stopped, and 30 mg daily dosing was chosen as the RP2D.
Within the phase II part of the study, the most common Grade 3–5 adverse events were lung infection (39%), febrile neutropenia (39%), other infections (29%), and sepsis (16%). A Grade 3–5 QTcF prolongation was recorded in one patient (3%). The most common Grade 1–2 adverse events included electrolyte abnormalities and elevated creatinine levels. Within the R/R AML cohort, with continuous dosing of quizartinib, the median time to achieve absolute neutrophil count (ANC) >500 was 51 days (n = 5). However, limiting the dose of quizartinib to 28 days per cycle reduced the median time to ANC >500 to 26 days.
Conclusion
The triplet combination of quizartinib, decitabine, and venetoclax has shown to be active in patients with FLT3-mutated R/R AML, including those previously exposed to FLT3 inhibitors, with a CR rate of 82%. The RP2D of quizartinib was 30 mg daily for this combination. RAS/MAPK and FLT3-F691L mutations were associated with resistance to treatment. This study is still ongoing.
References
Please indicate your level of agreement with the following statements:
The content was clear and easy to understand
The content addressed the learning objectives
The content was relevant to my practice
I will change my clinical practice as a result of this content


