All content on this site is intended for healthcare professionals only. By acknowledging this message and accessing the information on this website you are confirming that you are a healthcare professional. If you are a patient or carer, please visit Know AML.
The AML Hub website uses a third-party service provided by Google that dynamically translates web content. Translations are machine generated, so may not be an exact or complete translation, and the AML Hub cannot guarantee the accuracy of translated content. The AML Hub and its employees will not be liable for any direct, indirect, or consequential damages (even if foreseeable) resulting from use of the Google Translate feature. For further support with Google Translate, visit Google Translate Help.
The AML Hub is an independent medical education platform, sponsored by Daiichi Sankyo, Johnson & Johnson, Syndax, Thermo Fisher Scientific, Kura Oncology, and AbbVie. Funders are allowed no direct influence on our content. The levels of sponsorship listed are reflective of the amount of funding given. View funders.
Now you can support HCPs in making informed decisions for their patients
Your contribution helps us continuously deliver expertly curated content to HCPs worldwide. You will also have the opportunity to make a content suggestion for consideration and receive updates on the impact contributions are making to our content.
Find out more
Create an account and access these new features:
Bookmark content to read later
Select your specific areas of interest
View AML content recommended for you
Reduced-toxicity conditioning regimens: A comparison of irradiation vs chemotherapy for the treatment of AML
Pretransplant conditioning is vital in patients with acute myeloid leukemia (AML) receiving allogeneic hematopoietic cell transplantation (allo-HCT) to provide immunoablation and avoid posttransplant graft rejection. Sebastian Giebel and colleagues recently published in Bone Marrow Transplantation, a retrospective study on the optimal conditioning for AML patients receiving allo-HCT.1
The comparative study carried out by Giebel et al1 describes the impact of total body irradiation (TBI) at the dose of 8 Gy combined with fludarabine (TBI8Gy/Flu) or busulfan at the dose of 9.6 mg/kg (3 days) with fludarabine (Bu3/Flu), on patients with AML.
During this study, lower doses of TBI8Gy/Flu (30% reduction) and Bu3/Flu (25% reduction) were used compared with classical myeloablative regimens. Moreover, cyclophosphamide was substituted by fludarabine. The transplant conditioning intensity score was reported to be in intermediate range (2.5).
The two treatments were evaluated based on their reduced-toxicity conditioning for patients with AML treated with allo-HCT. The primary endpoint was leukemia-free survival (LFS), and secondary endpoints comprised: overall survival (OS), relapse incidence, nonrelapse mortality (NRM), incidence of Grades II–IV acute graft-versus-host disease (GvHD), incidence of chronic GvHD, and survival-free from Grades III–IV acute GvHD, chronic GvHD, and relapse (GRFS).
Study design and data collection
Data was collected from > 600 transplant centers associated with the European Society for Blood and Marrow Transplantation (EBMT), which contained information on adult patients in first remission post allo-HCT with an intermediate or high-risk karyotype, but who did not have ex vivo T-cell depletion. There were two study arms: Bu3/Flu, intravenous busulfan at a total dose of 9.6 mg/kg administered for 3 consecutive days with fludarabine (n = 350); and TBI8Gy/Flu, TBI at a total dose of 8 Gy in combination with fludarabine (n = 168).
Results
Median age for TBI8Gy/Flu patients was 50 years old, whereas the Bu3/Flu cohort contained patients in a significantly older age group. Also, the former group had a higher number of unrelated donors, as well as patients who received in vivo T-cell depletion. The median follow-up was 23 months for Bu3/Flu, and 57 months for TBI8Gy/Flu (Table 1).
Table 1. Patient, donor, and transplant-related characteristics and their significant differences1
|
Bu3/Flu, busulfan at the total dose of 9.6 mg/kg administered for 3 consecutive days + fludarabine; HLA, human leukocyte antigen; TBI8Gy/Flu, total body irradiation at a total dose of 8 Gy + fludarabine. |
|||
|
Characteristic |
Bu3/Flu (n = 350) |
TBI8Gy/Flu (n = 168) |
p value |
|---|---|---|---|
|
Median patient age, years (range) |
56.5 (18–73) |
50 (20.5–71) |
< 0.0001 |
|
Median interval from diagnosis to transplantation, months (range) |
4.9 (1.2–17.7) |
3.8 (1.8–15.9) |
< 0.0001 |
|
Median year of transplantation (range) |
2016 (2006–2018) |
2012 (2006–2018) |
<0.0001 |
|
Karnofsky performance status score ≥ 90%, % |
74 |
85 |
< 0.007 |
|
Donor type (HLA-identical), % |
47 |
58 |
0.02 |
|
In vivo T-cell depletion (Yes), % |
71 |
50 |
< 0.0001 |
|
Immunosuppression (Cyclosporin A alone), % |
27 |
2 |
< 0.0001 |
Univariate comparison (Figure 1) of Bu3/Flu and TBI8Gy/Flu depicted the following key findings:
- LFS rates were 66% for Bu3/Flu compared with 59.5% for TBI8Gy/Flu (p = 0.15)
- Occurrence of acute GvHD (Grade II–IV) was higher in Bu3/Flu than in the TBI8Gy/Flu cohort (9% vs 4%; p = 0.03)
- No significant difference could be detected in terms of the incidence of chronic GvHD and NRM for both study groups
- An increase in relapse incidence at 2 years was observed for Bu3/Flu compared with TBI8Gy/Flu (30% vs 20%; p = 0.01)
- OS rates were found to be 72.5% for TBI8Gy/Flu vs 62% for Bu3/Flu (p = 0.051)
Figure 1. Outcomes of univariate analysis for a all patients, b treatment group ≤ 50 y.o., and c treatment group ≥ 50 y.o.1
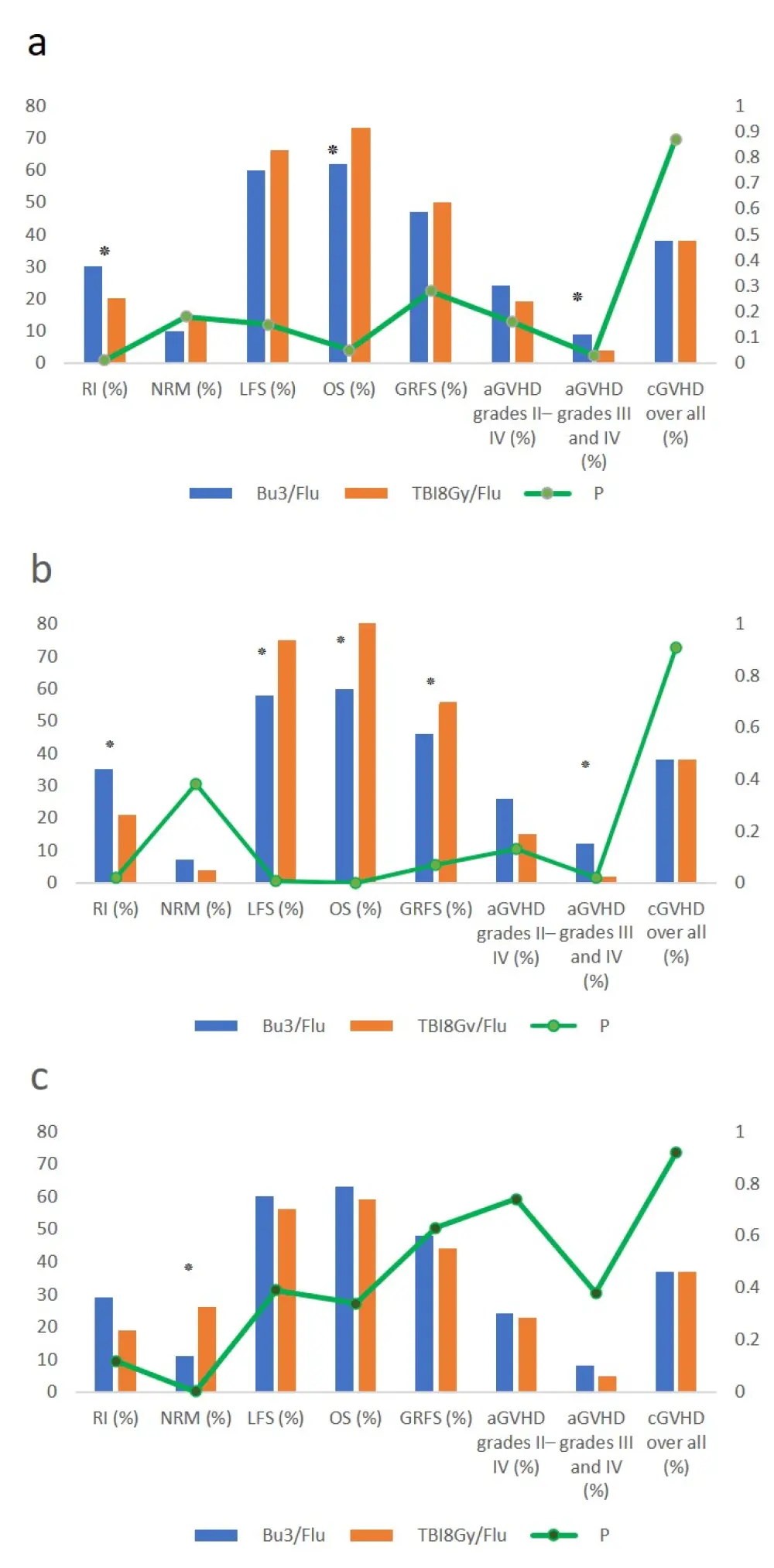
Data indicates a comparison between the BU3/Flu and TBI8Gy/Flu treatment groups.
*Marks significant difference in p values.
aGvHD, acute GvHD; cGvHD, chronic GvHD; GRFS, survival-free from relapse, acute GvHD Grades III and IV, and chronic GvHD; GvHD, graft-versus-host disease; LFS, leukemia-free survival; NRM, nonrelapse mortality; OS, overall survival; RI, relapse incidence; y.o., years old.
Interaction between age and type of conditioning
Separate analyses were performed stratifying patients by age. Comparison of Bu3/Flu with TBI8Gy/Flu revealed an increase for LFS (58% vs 75%; p = 0.007) and OS (60% vs 84%; p = 0.0005) in younger patients (< 50 years), as well as a reduction of Grades III and IV acute GvHD incidences (12% vs 2%; p = 0.02). However, in patients aged 50 years or older, the use of TBI8Gy/Flu was associated with an increased incidence of NRM (11% vs 26%; p = 0.002).
Similar results were observed during multivariate analysis. Overall, the TBI8Gy/Flu treatment proved to be more effective in patients under 50 years of age. Younger patients had an improved LFS rate (Hazard Ratio [HR] = 0.5; p = 0.04), OS (HR = 0.31; p = 0.004), GRFS (HR = 0.55; p = 0.03), and reduced risk of relapse (HR = 0.53; p = 0.08). On the contrary, the use of the same treatment in the older age group, resulted in an increased NRM (HR = 3.9; p = 0.0009). No other significant association between treatment regimens and other study points was recorded.
Conclusion
The study concluded that both reduced-toxicity treatments were associated with a low-risk of relapse and NRM, which could boost survival rates after allo-HCT. One of the limitations of this study was an unequal age group distribution of patients in both treatment groups. This may be the reason for the higher recovery rate and lower mortality rate observed in the TBI8Gy/Flu group. Nevertheless, TBI8Gy/Flu appears to be more effective in younger patients. In these patients, the increased efficacy brought about by high-dose homogeneity throughout the body may outweigh any early TBI toxicity, which diminishes over time. However, in patients older than 50 years, TBI toxicity may remain higher for longer, indicating the need for dose adjustments in these patients. Moreover, unbiased prospective studies are needed to assess the longer-term impact of these treatments.
References
Please indicate your level of agreement with the following statements:
The content was clear and easy to understand
The content addressed the learning objectives
The content was relevant to my practice
I will change my clinical practice as a result of this content
Your opinion matters
Which AML-related topic do you currently need the most practical guidance on?

