All content on this site is intended for healthcare professionals only. By acknowledging this message and accessing the information on this website you are confirming that you are a healthcare professional. If you are a patient or carer, please visit Know AML.
The AML Hub website uses a third-party service provided by Google that dynamically translates web content. Translations are machine generated, so may not be an exact or complete translation, and the AML Hub cannot guarantee the accuracy of translated content. The AML Hub and its employees will not be liable for any direct, indirect, or consequential damages (even if foreseeable) resulting from use of the Google Translate feature. For further support with Google Translate, visit Google Translate Help.
The AML Hub is an independent medical education platform, sponsored by Daiichi Sankyo, Johnson & Johnson, Syndax, Thermo Fisher Scientific, Kura Oncology, and AbbVie. Funders are allowed no direct influence on our content. The levels of sponsorship listed are reflective of the amount of funding given. View funders.
Now you can support HCPs in making informed decisions for their patients
Your contribution helps us continuously deliver expertly curated content to HCPs worldwide. You will also have the opportunity to make a content suggestion for consideration and receive updates on the impact contributions are making to our content.
Find out more
Create an account and access these new features:
Bookmark content to read later
Select your specific areas of interest
View AML content recommended for you
Reduced-toxicity conditioning regimen for older patients with AML: fludarabine plus treosulfan
Myeloablative conditioning regimen is associated with high non-relapse mortality (NRM) rates in older patients with acute myeloid leukemia (AML) or myelodysplastic syndromes (MDS).1 Regimens which fuse the lower organ toxicity associated with reduced intensity conditioning and the antileukemic activity observed with myeloablative conditioning have been provisionally named as reduced toxicity conditioning regimens.1 One promising reduced toxicity conditioning regimen is fludarabine + treosulfan (FluTreo).
Recently, Beelen et al.1 published an observational comparative analysis of FluTreo vs fludarabine + (FluMel) or + (BuCy) in Bone Marrow Transplantation. Here, we summarize the key findings below.
Methods1
- This was a retrospective European Society for Blood and Marrow Transplantation-registry based study.
- Data of patients from both the registry and the MC-FludT.14/L Trial II comparing FluTreo with FluBu (NCT00822393) was analyzed.
- A 1:1 propensity score matched analysis (PSA) was performed to reduce confounding due to differences between study and registry patients.
- The outcomes included a comparison of estimates of overall survival (OS), relapse incidence, and NRM within 2-years of transplant between the three conditioning regimens.
Key findings1
- A total of 968 and 287 patients with AML and MDS, respectively were included
- FluTreo (n = 174 and n = 78)
- FluMel (n = 256 and n = 82)
- BuCy (n = 503 and n = 127)
PSA outcome comparison in AML patients
- Figure 1 shows the 1:1 PSA for all three conditioning regimens.
- The 2-year NRM was lower for FluTreo vs FluMel in unpaired comparison (p = 0.019).
- The 2-year OS was higher for FluTreo vs FluMel in unpaired comparison (p = 0.04).
- The 2-year OS was higher for FluTreo vs BuCy and was significantly different in both paired (p < 0.001) and unpaired comparison (p < 0.001).
Figure 1. 1:1 PSA of clinical endpoints 2-years posttransplant for A FluTreo vs FluMel and B FluTreo vs BuCy*
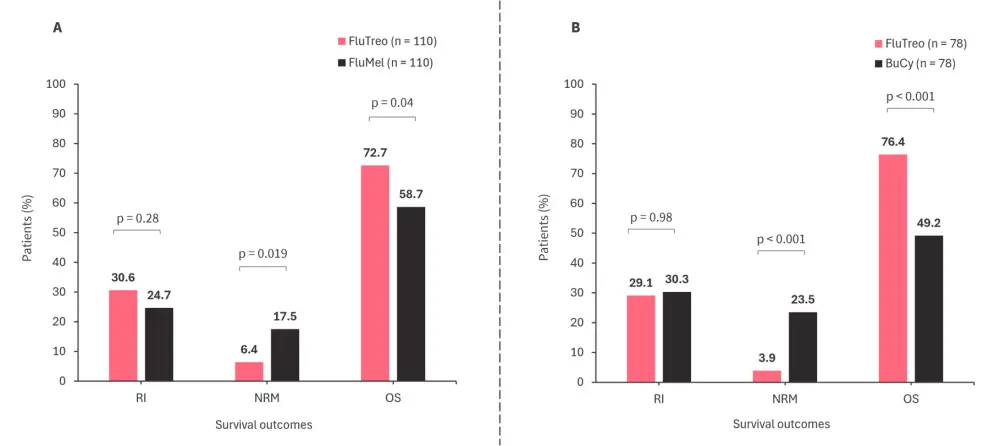
BuCy, busulfan + cyclophosphamide; FluMel, fludarabine + melphalan; FluTreo, fludarabine + treosulfan; NRM, non-relapse mortality; OS, overall survival; PSA, propensity score matched analysis; RI, relapse incidence.
*Adapted from Beelen, et al.1
PSA outcome comparison in MDS patients
- The 2-year NRM was lower for FluTreo vs BuCy in both paired and unpaired comparison (p = 0.13 and p = 0.18).
- The 2-year OS was higher for FluTreo vs BuCy in both paired and unpaired comparison (p = 0.01 each).
Multivariate comparison of outcomes
- For AML patients, multivariate analysis corroborated all significant results obtained by PSA for 2-year NRM and OS endpoints.
- For MDS patients, results of 2-year NRM and OS endpoints were also confirmed.
- Only 2-year OS between FluTreo and BuCy regimens was significantly different.
|
Key learnings |
|---|
|
References
Please indicate your level of agreement with the following statements:
The content was clear and easy to understand
The content addressed the learning objectives
The content was relevant to my practice
I will change my clinical practice as a result of this content

