All content on this site is intended for healthcare professionals only. By acknowledging this message and accessing the information on this website you are confirming that you are a healthcare professional. If you are a patient or carer, please visit Know AML.
The AML Hub website uses a third-party service provided by Google that dynamically translates web content. Translations are machine generated, so may not be an exact or complete translation, and the AML Hub cannot guarantee the accuracy of translated content. The AML Hub and its employees will not be liable for any direct, indirect, or consequential damages (even if foreseeable) resulting from use of the Google Translate feature. For further support with Google Translate, visit Google Translate Help.
The AML Hub is an independent medical education platform, sponsored by Daiichi Sankyo, Johnson & Johnson, Syndax, Thermo Fisher Scientific, Kura Oncology, and AbbVie. Funders are allowed no direct influence on our content. The levels of sponsorship listed are reflective of the amount of funding given. View funders.
Now you can support HCPs in making informed decisions for their patients
Your contribution helps us continuously deliver expertly curated content to HCPs worldwide. You will also have the opportunity to make a content suggestion for consideration and receive updates on the impact contributions are making to our content.
Find out more
Create an account to access:
Bookmark & personalize site content
Receive alerts for new content in your areas of interest
View AML content recommended for you
Efficacy and safety of midostaurin in younger and older patients with FLT3-ITD AML
Midostaurin is a FLT3-ITD inhibitor approved by the U.S. Food and Drug Administration (FDA) and European Commission (EC) for newly diagnosed AML in adult patients with FLT3-mutated AML. This decision followed results from the RATIFY trial which demonstrated midostaurin and intensive chemotherapy improved event-free survival (EFS) and overall survival (OS) compared with placebo. In the recent phase II AMLSG 16-10 trial (NCT01477606), older patients aged 61–70 years old were included in the treatment cohort with midostaurin. Interim study results have been previously reported by the AML Hub. Here, we summarize the key efficacy and safety results, published by Döhner et al.1, in Blood Advances.
Methods
The AMLSG 16-10 was a single arm, phase II study1 conducted at 54 centres in Austria and Germany. The outcomes of patients enrolled in this trial were compared with those of a historical control cohort (AMLSG). Patient inclusion criteria and study endpoints are shown in Figure 1.
Figure 1. Overview of the phase II AMLSG 16-10 trial*
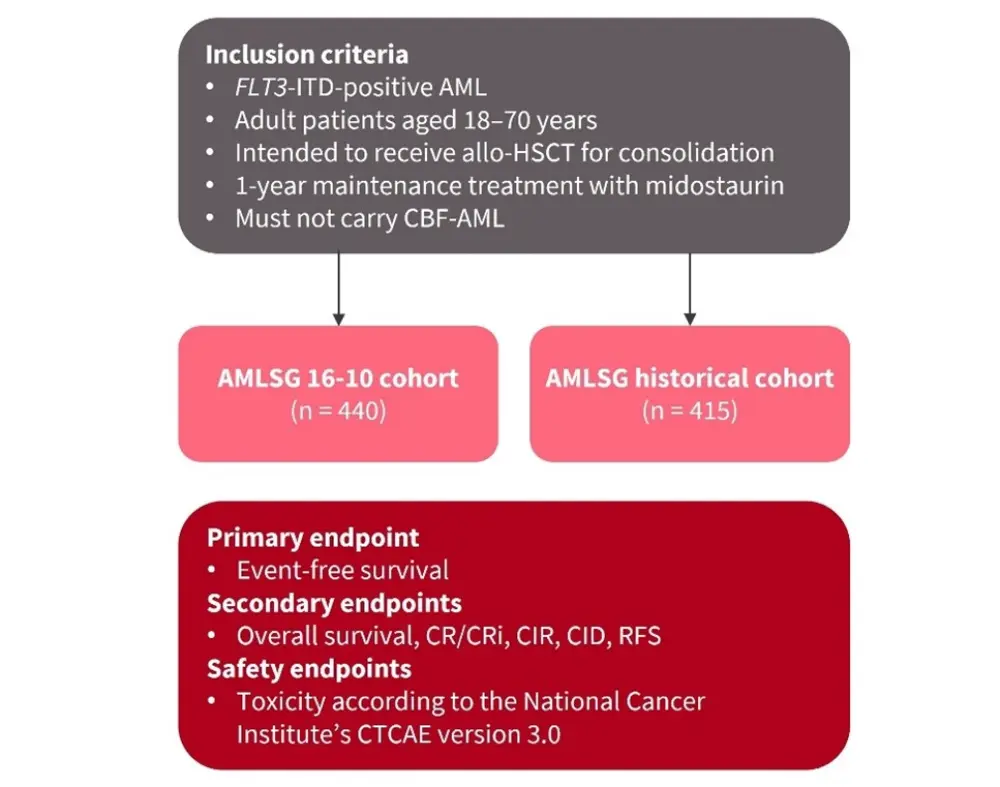
Allo-HSCT, allogeneic hematopoietic stem cell transplant; AML, acute myeloid leukemia; CBF-AML, core-binding factor AML; CID, cumulative incidence of death; CIR, cumulative incidence of relapse; CR/CRi, complete remission/complete remission with incomplete hematologic recovery; CTCAE, Common Terminology Criteria for Adverse Events; RFS, relapse-free survival.
*Adapted from Döhner et al.1
Results
Patient characteristics for the AMLSG 16-10 and AMLSG historical cohort are shown in Table 1.
Table 1. Patient characteristics*
|
BM, bone marrow; Hb, hemoglobin; ECOG, Eastern Cooperative Oncology Group; PB, peripheral blood; WBC, white blood cell. |
|||
|
Characteristic |
AMLSG 16-10 cohort |
AMLSG historical cohort |
p value |
|---|---|---|---|
|
Median age, years (range) |
54.1 (18–70) |
50.5 (18–70) |
< 0.001 |
|
Female, % |
57 |
54 |
|
|
ECOG PS, % |
< 0.0001 |
||
|
0 |
38 |
22 |
|
|
1 |
50 |
61 |
|
|
2 |
12 |
16 |
|
|
WBC, × 109/L |
|||
|
Median (range) |
41.8 (0.3–420) |
44.8 (0.2–439) |
|
|
Missing, n |
3 |
3 |
|
|
Hb, g/dL |
|||
|
Median (range) |
9.0 (4.1–18.1) |
9.0 (3.1–16.6) |
|
|
Missing, n |
4 |
3 |
|
|
Platelet count, × 109/L |
|||
|
Median (range) |
59 (5–681) |
58 (6–734) |
|
|
Missing, n |
3 |
2 |
|
|
BM blasts, % |
|||
|
Median (range) |
80 (0–100) |
85 (2–100) |
|
|
Missing, n |
46 |
25 |
|
|
PB blasts, % |
|||
|
Median (range) |
52 (0–100) |
60 (0–100) |
|
|
Missing, n |
30 |
20 |
|
|
AML type, % |
< 0.0001 |
||
|
De novo |
89 |
91 |
|
|
Secondary |
7 |
1 |
|
|
Therapy-related |
4 |
3 |
|
|
Missing, n |
— |
1 |
|
|
Cytogenetics, % |
0.02 |
||
|
Intermediate-I |
69 |
78 |
|
|
Intermediate-II |
25 |
17 |
|
|
Adverse |
6 |
5 |
|
|
Missing, n |
28 |
0 |
|
|
FLT3-ITD, % |
|||
|
Allelic ratio ≥0.5 |
55 |
56 |
|
|
Missing, n |
2 |
121 |
|
|
FLT3-TKD present, % |
4 |
4 |
|
|
Missing, n |
— |
22 |
|
Efficacy
- The median follow-up for the AMLSG 16-10 and historical cohort was 40.4 months and 76.3 months, respectively
- The 2-year EFS and OS were greater in the AMLSG 16-10 cohort treated with midostaurin compared to the historical cohort (Figure 2)
- The AMLSG 16-10 cohort also showed improved EFS and OS when assessing via subgroup analysis for younger (18–60 years) and older (61–70) patients
Figure 2. 2-year EFS and OS in the AMLSG 16-10 and AMLSG historical cohort*
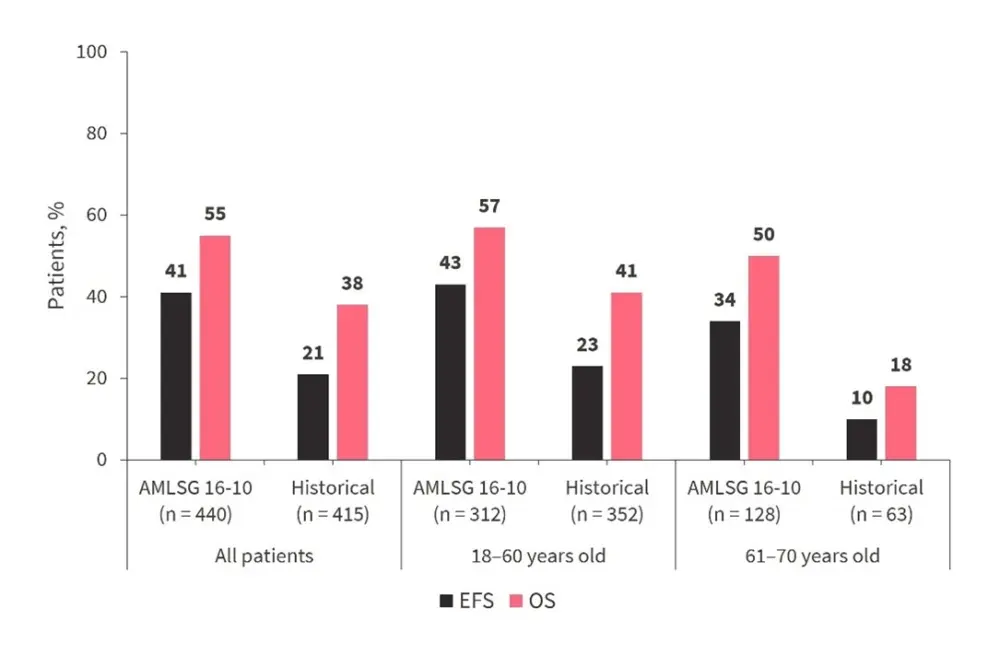
*Adapted from Döhner et al.1
- In a multivariate analysis, midostaurin treatment in the AMLSG 16-10 trial was associated with significantly improved OS and EFS compared with the historical cohort (Table 2)
- This effect was again observed in both younger and older patients.
Table 2. Multivariate analysis*
|
CI, confidence interval; EFS, event-free survival; HR, hazard ratio; OS, overall survival; WBC, white blood cell. |
||||
|
Variable |
EFS |
OS |
||
|---|---|---|---|---|
|
HR (95% CI) |
p value |
HR (95% CI) |
p value |
|
|
All patients (N = 855) |
||||
|
AMLSG 16-10 |
0.55 |
<0.001 |
0.56 |
<0.001 |
|
Age (per 10 years) |
1.17 |
<0.001 |
1.33 |
<0.001 |
|
NPM1-mutated |
0.48 |
<0.001 |
0.76 |
0.002 |
|
WBC |
1.21 |
0.011 |
1.23 |
0.015 |
|
18–60 years (n = 664) |
||||
|
AMLSG 16-10 |
0.59 |
<0.001 |
0.59 |
<0.001 |
|
Age (per 10 years) |
1.16 |
<0.001 |
1.30 |
<0.001 |
|
NPM1-mutated |
0.47 |
<0.001 |
0.76 |
0.010 |
|
WBC |
1.24 |
0.014 |
1.23 |
0.037 |
|
61–70 years (n = 191) |
||||
|
AMLSG 16-10 |
0.41 |
<0.001 |
0.47 |
<0.001 |
|
Female sex |
— |
— |
0.70 |
0.042 |
|
NPM1-mutated |
0.53 |
<0.001 |
— |
— |
- In a sensitivity analysis, allogeneic hematopoietic stem cell transplantation (allo-HSCT) slightly reduced the treatment effect compared to the multivariate model without considering allo-HSCT (EFS: HR 0.63 and 0.62 vs 0.55; OS: HR 0.72 and 0.66 vs 0.57)
- When comparing patients from AMLSG 16-10 with the RATIFY trial, the treatment effect of midostaurin remained significant (HR, 0.71; p = 0.005)
- Treatment with midostaurin produced significantly greater complete remission/complete remission with incomplete hematologic recovery (CR/CRi) compared with the historical cohort, and lower cumulative incidence of relapse (CIR) and rates of refractory disease
- The 2-year RFS rate in the AMLSG 16-10 trial was 52% and in the historical population was 32%, and multivariate analysis revealed treatment within the AMLSG 16-10 trial (HR, 0.50; p < 0.001) and NPM1 mutation (HR, 0.63; P < 0.001) as significant favorable factors, while older age (HR per 10 years, 1.02; p < 0.001) was an adverse factor.
Table 3. Analysis of secondary endpoints*
|
CID, cumulative incidence of death; CIR, cumulative incidence of relapse; CR/CRi, complete remission/complete remission with incomplete hematologic recovery; ED, early death; HD, hypoplastic death; RD, refractory disease. |
||||||
|
Outcomes, % (unless otherwise stated) |
AMLSG 16-10 cohort |
Historical cohort |
||||
|---|---|---|---|---|---|---|
|
All patients |
18–60 years |
61–70 years |
All patients |
18–60 years |
61–70 years |
|
|
CR/CRi |
74.9 |
75.9 |
72.4 |
64.6 |
66.5 |
54.0 |
|
CIR |
28.0 |
24.0 |
37.0 |
57.0 |
54.0 |
74.0 |
|
CID |
20.0 |
20.0 |
19.0 |
12.0 |
12.0 |
12.0 |
|
RD |
19.2 |
20.6 |
15.8 |
30.6 |
28.4 |
42.9 |
|
ED/HD |
5.9 |
3.5 |
11.8 |
4.8 |
5.1 |
3.2 |
|
Missing, n |
2 |
1 |
1 |
— |
— |
— |
Safety
- Midostaurin treatment was interrupted at least once in 40% of patients, mostly due to toxicity (73%)
- The most frequent adverse events (AEs) leading to treatment discontinuation were thrombocytopenia, nausea/vomiting, graft-versus-host disease (GvHD), hepatobiliary disease/increase of transaminases, and infections
- Grade ≥3 AEs are summarized in Table 4.
- Metabolism and nutrition disorders (p = 0.003), vascular disorders (p = 0.04), cardiac disorders (p = 0.08), and respiratory, thoracic, and mediastinal disorders (p = 0.07) were more common in older (61–70 years) patients
Table 4. Grade ≥3 AEs occurring in >10% of patients*
|
AEs, adverse events. |
|
|
AEs, % |
Patients (N = 440) |
|---|---|
|
Blood and lymphatic system disorders |
95 |
|
Infections and infestations |
66 |
|
Gastrointestinal |
39 |
|
General disorders |
34 |
|
Investigations |
30 |
|
Metabolism and nutrition disorders |
27 |
|
Respiratory, thoracic, and mediastinal |
17 |
|
Vascular |
15 |
|
Renal and urinary |
12 |
|
Nervous system |
10 |
|
Cardiac |
10 |
|
Skin and subcutaneous tissue |
10 |
Conclusion
Results from the phase II study1 showed the benefits of midostaurin in patients with FLT3-ITD-mutant AML. In line with the previous RATIFY trial, compared to historical controls, treatment with midostaurin significantly improved the survival outcomes in older and younger patients with FLT3-ITD-positive AML with a manageable safety profile.
References
Please indicate your level of agreement with the following statements:
The content was clear and easy to understand
The content addressed the learning objectives
The content was relevant to my practice
I will change my clinical practice as a result of this content
Your opinion matters
Which AML-related topic do you currently need the most practical guidance on?

