All content on this site is intended for healthcare professionals only. By acknowledging this message and accessing the information on this website you are confirming that you are a healthcare professional. If you are a patient or carer, please visit Know AML.
The AML Hub website uses a third-party service provided by Google that dynamically translates web content. Translations are machine generated, so may not be an exact or complete translation, and the AML Hub cannot guarantee the accuracy of translated content. The AML Hub and its employees will not be liable for any direct, indirect, or consequential damages (even if foreseeable) resulting from use of the Google Translate feature. For further support with Google Translate, visit Google Translate Help.
The AML Hub is an independent medical education platform, sponsored by Daiichi Sankyo, Johnson & Johnson, Syndax, Thermo Fisher Scientific, Kura Oncology, and AbbVie. Funders are allowed no direct influence on our content. The levels of sponsorship listed are reflective of the amount of funding given. View funders.
Now you can support HCPs in making informed decisions for their patients
Your contribution helps us continuously deliver expertly curated content to HCPs worldwide. You will also have the opportunity to make a content suggestion for consideration and receive updates on the impact contributions are making to our content.
Find out more
Create an account to access:
Bookmark & personalize site content
Receive alerts for new content in your areas of interest
View AML content recommended for you
ASH 2022 roundup: IDH inhibitors
Approximately 20% of patients with acute myeloid leukemia (AML) are found to have somatic mutations in genes encoding the isocitrate dehydrogenase (IDH) enzyme, with ~5–10% of patients having an IDH1 mutation and ~15% of patients having an IDH2 mutation.1,2 Previous reports have suggested that patients with IDH-mutated AML have a higher 1-year relapse rate after allogeneic hematopoietic stem cell transplantation (allo-HSCT) compared with patients without IDH mutations.1 Here, we discuss four presentations from the 64th American Society of Hematology (ASH) Annual Meeting and Exposition; the speakers discussed relapse rates in patients with IDH mutations and the use of enasidenib and ivosidenib treatment in this patient population.
Prognostic value of MRD in IDH mutated-AML3
Among patients in complete remission (CR), measurable residual disease (MRD) prior to allo-HSCT has been shown to increase posttransplant relapse risk. In a study presented by Ravindra,3 which included 56 patients with IDH-mutated AML who received an allo-HSCT at the Johns Hopkins Sidney Kimmel Comprehensive Cancer Center, US, between 2015 and 2020, pre- and posttransplant bone marrow samples were sequenced using droplet digital polymerase chain reaction or next generation sequencing (NGS); of these patients, 28, 26, and 2 patients had IDH1, IDH2, or both mutations.
Key findings
Pretransplant droplet digital polymerase chain reaction/NGS analysis (n = 50) showed 56% of patients with an IDH mutation were also MRD-positive. Posttransplant (n = 48), 10% of patients were MRD positive; two of these patients subsequently experienced disease progression and died, whereas three remained in remission. The original IDH mutation was present at relapse (n = 10) in 90% of patients, with additional mutations also found in several samples. Overall survival (OS) was similar between patients with persistent mutations in IDH1 or IDH2 and those without. In addition, CR rates were comparable between patients with MRD-positivity and MRD-negativity, irrespective of IDH-mutation status.
In this study, the presence of pre- or posttransplant MRD in patients with IDH mutations was not found to be prognostic of relapse.
Enasidenib
Posttransplant enasidenib maintenance1
Salhotra discussed a multicenter, pilot phase II trial (NCT03728335) in 15 adult patients with IDH2-mutated AML who received enasidenib as post-HSCT maintenance therapy. The primary objective was safety and tolerability, with secondary objectives including OS, leukemia-free survival, non-relapse mortality, and 1-year chronic graft-versus-host disease-free and relapse-free survival. Baseline patient characteristics are shown in Table 1.
Table 1. Baseline patient characteristics*
|
CR, complete remission; HSCT, hematopoietic stem cell transplant; MAC, myeloablative conditioning; MRD, measurable residual disease; NMA, non-myeloablative; RIC, reduced-intensity conditioning. |
|
|
Characteristic, % (unless otherwise stated) |
N = 15 |
|---|---|
|
Median age (range), years |
58 (27–77) |
|
Gender ratio, male:female |
6:9 |
|
Median time to start enasidenib post-HSCT (range), days |
104 (54–131) |
|
Disease status at transplant |
|
|
First CR |
66 |
|
Second CR |
26 |
|
MRD positivity |
7 |
|
Cytogenetics |
|
|
Favorable |
13.3 |
|
Intermediate |
60 |
|
Adverse |
26 |
|
Conditioning treatment |
|
|
NMA/RIC |
53 |
|
MAC |
47 |
The most common Grade 1–3 adverse events in Cycle 1 and 2 of maintenance therapy were anemia, nausea, hyperbilirubinemia, and lymphopenia.1 The incidence of adverse events was similar from Cycle 3 onwards, with the most common Grade 1–3 adverse events being anemia, diarrhea, hyperbilirubinemia, and neutropenia. At data cut-off (June 2022), 33% of patients had completed treatment; the median number of completed cycles was 20 cycles (range, 1–24 cycles). In this trial, enasidenib was well tolerated and effectively prevented relapse in patients with IDH2 mutations. The Kaplan-Meier estimate for leukemia-free survival was 1.0 at both 12 and 24 months, with an estimate of 0.8 for chronic graft-versus-host disease-free relapse-free survival at 12 and 24 months.
Achievement of complete remission with enasidenib4
The updated results and 3-year follow-up of the Beat AML Master Study phase II/Ib sub-study (NCT03013998) were presented by Cai.4 The design of the sub-study, which included patients ≥60 years of age with R140/R172 mutations in IDH2, is shown in Figure 1.
Figure 1. Beat AML Master Study phase II/Ib sub-study design*
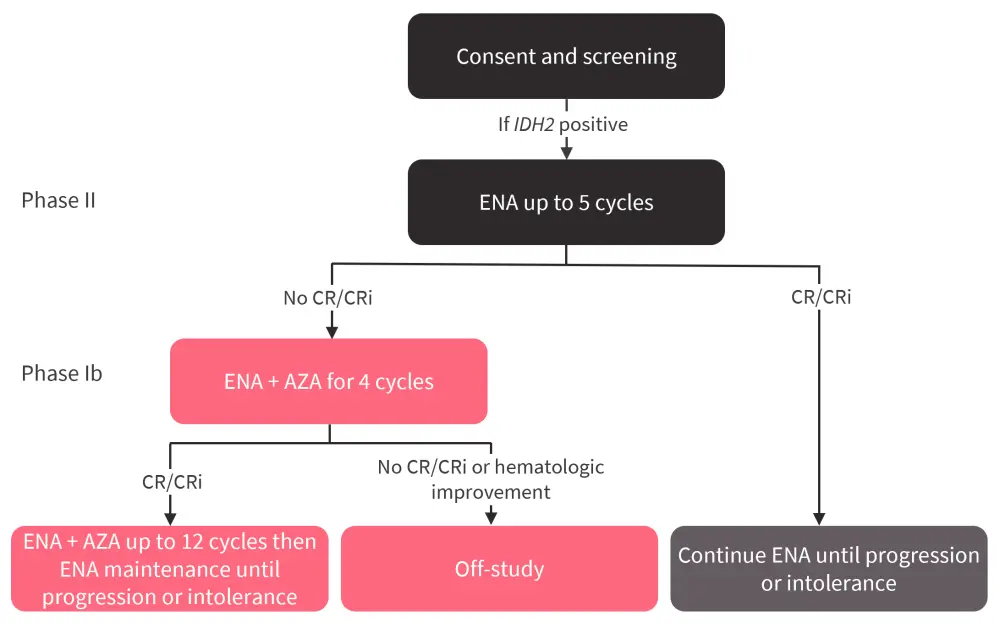
AZA, azacitidine; CR, complete remission; CRi, CR with incomplete hematologic recovery; ENA, enasidenib; IDH2, isocitrate dehydrogenase 2.
*Adapted from Cai.4
The primary endpoint of the sub-study was CR or CR with incomplete hematologic recovery in patients treated with enasidenib. The baseline patient characteristics are shown in Table 2.
Table 2. Baseline characteristics of patients in the Beat AML Master Study phase II/Ib sub-study*
|
ECOG, Eastern Cooperative Oncology Group; ELN, European Leukemia Net; IDH2, isocitrate dehydrogenase 2. |
||
|
Characteristic, % (unless |
Phase II (enasidenib |
Phase Ib (enasidenib + azacitidine) |
|---|---|---|
|
Median age (range), years |
75 (60–89) |
75 (60–86) |
|
Gender |
|
|
|
Male |
48 |
59 |
|
Female |
52 |
41 |
|
ECOG Performance Status |
|
|
|
0 |
27 |
41 |
|
1 |
53 |
35 |
|
2 |
20 |
24 |
|
ELN risk |
|
|
|
Adverse |
53 |
47 |
|
Intermediate |
28 |
29 |
|
Favorable |
18 |
24 |
|
Mutated IDH2 |
|
|
|
Median (range), n |
40.5 (6–58) |
36 (14–51) |
|
R140 |
73 |
47 |
|
R172 |
27 |
53 |
Median duration of response was 11.1 months and 14.6 months for phase II and phase Ib, respectively, with a median OS of 17.1 months and 12.5 months, respectively.4 Nine patients harbored TP53 mutations (usually patients with TP53 mutations are assigned to other Beat AML sub-studies) and TP53 mutations did not appear to impact CR or CR with incomplete hematologic recovery response. Response rates and the incidence of disease progression in both study phases are shown in Figure 2.
Figure 2. Response and progression rates*
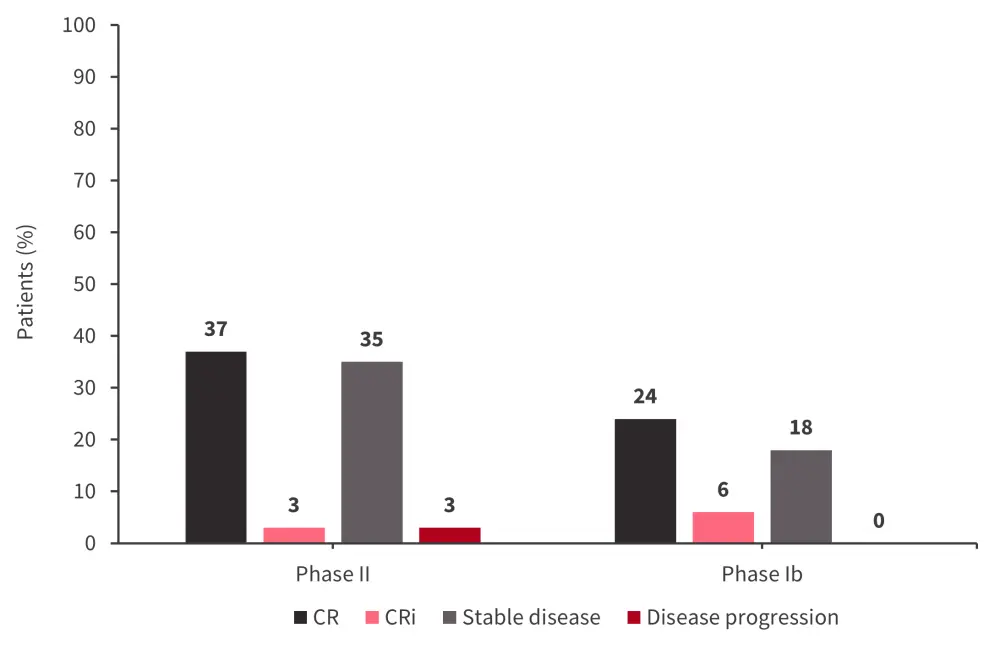
CR, complete remission; CRi, CR with incomplete hematologic recovery.
*Adapted from Cai.4
The most common adverse events were,
- in phase II: nausea (58.3%), anemia (56.7%), hypokalemia (56.7%), diarrhea (43.3%), musculoskeletal pain (43.3%), and decreased appetite (40%); and
- in phase Ib: anemia (41.2%), nausea (35.3%), vomiting (35.3%), hypoalbuminemia (35.3%), hyponatremia (35.3%), and hypophosphatemia (35.3%).
Differentiation syndrome was also common in both the phase II (23.3%) and Ib (17.6%), with tumor lysis syndrome occurring in 3.3% of patients in phase II and no patients in phase Ib.
This study demonstrated that enasidenib can be used safely and effectively in patients with IDH2-mutated AML. However, addition of azacitidine may reduce toxicities whilst retaining the efficacy of enasidenib monotherapy.
Mutational clearance with ivosidenib2
Ivosidenib, a selective IDH1 enzyme inhibitor, was approved in 2022 by the U.S. Food and Drug Administration (FDA) for the treatment of patients with relapsed/refractory IDH1-mutated AML in combination with azacitidine. Dohner presented an update from the AGILE study (NCT03173248), which compared outcomes of ivosidenib plus azacitidine versus placebo plus azacitidine in patients with newly diagnosed IDH1-mutated AML and discussed the clearance of co-mutations and the emergence of mutations leading to relapse.
Of the 146 patients with newly diagnosed AML (ivosidenib plus azacitidine, n = 76; placebo plus azacitidine, n = 74), 46 who received ivosidenib plus azacitidine had molecular data available; of these, eight had NGS data and seven had single-cell DNA sequencing data available at the time of relapse.
Key findings
An association was found between achievement of CR and clearance of baseline mutations, with 73% of patients who achieved CR after treatment with ivosidenib plus azacitidine (n = 30) also clearing all baseline mutations at ≥1 on-study timepoint. In total, 30 of 33 patients who achieved CR/CRi and harbored an IDH1 mutation demonstrated mutational clearance, this was not found to be associated with baseline variant allele frequency.
Mutations emerging whilst on treatment were identified in 22 of 46 patients, including seven patients who had achieved CR and cleared all baseline mutations. However, no second-site IDH1 mutations were noted, although an emerging IDH2 mutation was found in two patients. Relapse occurred in six of eight patients, with mutations found in the following genes:
- Patient 1: PHF6, RUNX1, TET2
- Patient 2: EZH2, JAK2, ZRSR2
- Patient 3: ASXL1, FLT3-ITD, KRAS
- Patient 4: ASXL1, TET2
- Patient 5: TP53
- Patient 6: CREBBP, FLT3-TKD, RUNX1
This study demonstrated that use of ivosidenib and azacitidine led to durable responses and clearance of baseline IDH1 mutations. Mutated IDH1 was not detected at relapse in six of eight patients who experienced disease progression.
Conclusion
In the study presented by Ravindra,3 NGS revealed similar OS between patients with persistent IDH1 or IDH2 mutations and those without and the presence of pre- or posttransplant MRD in patients with IDH mutations was not found to be prognostic of relapse. However, this study was limited by the small cohort size.3
In both studies, enasidenib was found to be well tolerated; the most common adverse events were anemia and gastrointestinal events.1,4 The Beat AML study also demonstrated similar efficacy with enasidenib in combination with azacitidine compared with enasidenib monotherapy, and there were also fewer toxicities.4 Treatment with ivosidenib in combination with azacitidine achieved higher CR rates and cleared baseline IDH mutations.2 Future studies of enasidenib and ivosidenib with larger cohort sizes are warranted to provide further data on the efficacy of these treatments in patients with IDH-mutated AML.
References
Please indicate your level of agreement with the following statements:
The content was clear and easy to understand
The content addressed the learning objectives
The content was relevant to my practice
I will change my clinical practice as a result of this content
Your opinion matters
Which AML-related topic do you currently need the most practical guidance on?

