All content on this site is intended for healthcare professionals only. By acknowledging this message and accessing the information on this website you are confirming that you are a healthcare professional. If you are a patient or carer, please visit Know AML.
The AML Hub website uses a third-party service provided by Google that dynamically translates web content. Translations are machine generated, so may not be an exact or complete translation, and the AML Hub cannot guarantee the accuracy of translated content. The AML Hub and its employees will not be liable for any direct, indirect, or consequential damages (even if foreseeable) resulting from use of the Google Translate feature. For further support with Google Translate, visit Google Translate Help.
The AML Hub is an independent medical education platform, sponsored by Daiichi Sankyo, Johnson & Johnson, Syndax, Thermo Fisher Scientific, Kura Oncology, and AbbVie. Funders are allowed no direct influence on our content. The levels of sponsorship listed are reflective of the amount of funding given. View funders.
Now you can support HCPs in making informed decisions for their patients
Your contribution helps us continuously deliver expertly curated content to HCPs worldwide. You will also have the opportunity to make a content suggestion for consideration and receive updates on the impact contributions are making to our content.
Find out more
Create an account and access these new features:
Bookmark content to read later
Select your specific areas of interest
View AML content recommended for you
Visual abstract | Phase I/II trial of SAR443579 in patients with R/R AML, B-ALL, or HR-MDS
Featured:
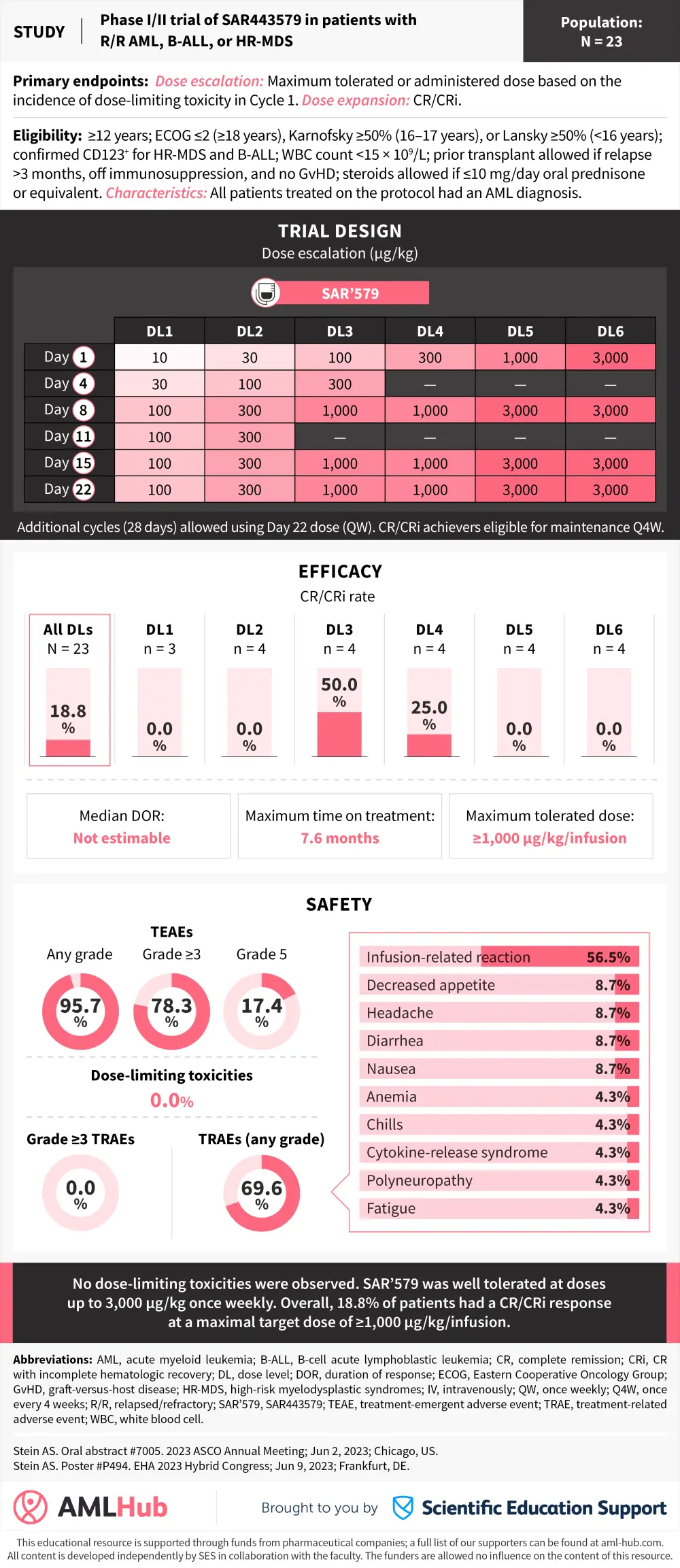
The AML Hub is pleased to present a visual abstract summarizing the preliminary safety and efficacy results from the phase I/II trial (NCT05086315) investigating SAR443579 (SAR’579), a trifunctional anti-CD123 NKp46×CD16 natural killer cell engager, in patients with relapsed/refractory acute myeloid leukemia.1,2
Preliminary results from this trial were presented by Stein1,2 at the 2023 American Society of Clinical Oncology (ASCO) Annual Meeting and European Hematology Association (EHA) 2023 Hybrid Congress. No dose-limiting toxicities were identified. Based on the results of this analysis, the study protocol was amended to assess a higher dose of SAR’579.1,2
References
Please indicate your level of agreement with the following statements:
The content was clear and easy to understand
The content addressed the learning objectives
The content was relevant to my practice
I will change my clinical practice as a result of this content
Your opinion matters
Which AML-related topic do you currently need the most practical guidance on?


 Suzanna de Barra
Suzanna de Barra Dylan Barrett
Dylan Barrett