All content on this site is intended for healthcare professionals only. By acknowledging this message and accessing the information on this website you are confirming that you are a healthcare professional. If you are a patient or carer, please visit Know AML.
The AML Hub website uses a third-party service provided by Google that dynamically translates web content. Translations are machine generated, so may not be an exact or complete translation, and the AML Hub cannot guarantee the accuracy of translated content. The AML Hub and its employees will not be liable for any direct, indirect, or consequential damages (even if foreseeable) resulting from use of the Google Translate feature. For further support with Google Translate, visit Google Translate Help.
The AML Hub is an independent medical education platform, sponsored by Daiichi Sankyo, Johnson & Johnson, Syndax, Thermo Fisher Scientific, Kura Oncology, and AbbVie. Funders are allowed no direct influence on our content. The levels of sponsorship listed are reflective of the amount of funding given. View funders.
Now you can support HCPs in making informed decisions for their patients
Your contribution helps us continuously deliver expertly curated content to HCPs worldwide. You will also have the opportunity to make a content suggestion for consideration and receive updates on the impact contributions are making to our content.
Find out more
Create an account and access these new features:
Bookmark content to read later
Select your specific areas of interest
View AML content recommended for you
Validation of the ELN 2017 risk stratification for acute myeloid leukemia
The European LeukemiaNet (ELN) 2017 risk classification is considered an essential guide for prognosis assessment in acute myeloid leukemia (AML) and to inform treatment decisions after patients achieve remission; however, only a few studies have validated its prognostic value in large patient populations.
Alex Bataller and colleagues1 have attempted to validate the ELN 2017 risk classification in patients with AML who were treated according to risk-adapted post remission therapy within the Spanish AML cooperative group Cooperativo Para el Estudio y Tratamiento de las Leucemias Agudas y Mielodisplasias (CETLAM) protocol (CETLAM-12). Patients were retrospectively assigned to the corresponding ELN 2017 risk category and treatment outcomes were analyzed. The results were recently published in Blood Advances.1
CETLAM-12 treatment protocol is established for untreated, fit patients up to 70 years diagnosed with de novo AML and uses a similar risk stratification to the ELN 2017; favorable risk characteristics are the same and there are a few differences in intermediate and adverse-risk groups (Table 1).
Table 1. Comparison of AML risk classifications by the ELN 2017 and CETLAM-12
|
CETLAM, Cooperativo Para el Estudio y Tratamiento de las Leucemias Agudas y Mielodisplasias; ELN 2017, The European LeukemiaNet 2017 risk classification |
||
|
|
ELN 2017 |
CETLAM-12 |
|---|---|---|
|
Favorable risk |
t(8;21)(q22;q22); RUNX1-RUNX1T1 |
|
|
inv(16)(p13.1q22) or t(16;16)(p13.1;q22); CBFB-MYH11 |
||
|
Biallelic mutated CEBPA |
||
|
Mutated NPM1 without FLT3-ITD or with FLT3-ITDlow |
||
|
Intermediate-I risk |
Mutated NPM1 and FLT3-ITDhigh |
— |
|
Wild-type NPM1 without FLT3-ITD or with FLT3-ITDlow (without adverse-risk genetic lesions) |
— |
|
|
Intermediate-II risk |
t(9;11)(p21.3;q23.3); MLLT3-KMT2A |
— |
|
Cytogenetic abnormalities not classified as favorable or adverse |
Cytogenetic abnormalities not classified as favorable or adverse |
|
|
Adverse risk |
t(v;11q23.3); KMT2A rearranged |
|
|
t(6;9)(p23;q34.1); DEK-NUP214 |
||
|
inv(3)(q21.3q26.2) or t(3;3)(q21.3;q26.2); GATA2,MECOM (EVI1) |
abn(3q) excluding t(3;5)(q21-25;q31-35); inv(3)(q21q26)/t(3;3)(q21;q26) |
|
|
5 or del(5q); -7; -17/abn(17p); Complex karyotype, monosomal karyotype
|
add(5q),del(5q),-5, -7, add(7q)/del(7q), 17/abn(17p); Complex karyotype, monosomal karyotype
|
|
|
t(9;22)(q34.1;q11.2); BCR-ABL1 |
t(9;22)(q34;q11) ; BCR-ABL1 |
|
|
Wild-type NPM1 and FLT3-ITDhigh
|
FLT3-ITDhigh with or without NPM1 mutation |
|
|
Mutated RUNX1, ASXL1 or TP53
|
— |
|
The risk-adapted post-remission strategy (following a 3 + 7 induction regimen with idarubicin and cytarabine) in the CETLAM-12 protocol is summarized below:
- Favorable risk group
- Consolidation therapy: 3 courses of HDAC
- BM MRD assessment: After chemotherapy course and thereafter at 3-month intervals for at least 3 years after CR
- Allogeneic stem cell transplantation (allo-SCT): in case of a confirmed molecular failure
- Transplant-ineligible patients: HMA or clinical trial
- Intermediate and adverse-risk groups
- Consolidation therapy: At least 1 course of HDAC
- Patients with persistent minimal residual disease (MRD) post-consolidation allocated to high-risk category and allo-SCT was recommended.
- Patients with FLT3 mutations could receive midostaurin within an early access program
Visual abstract
Below, we provide a visual abstract to summarize key outcomes by the ELN 2017 risk classification as well as a subset of patients with a poor prognosis within the ELN 2017 adverse-risk group.
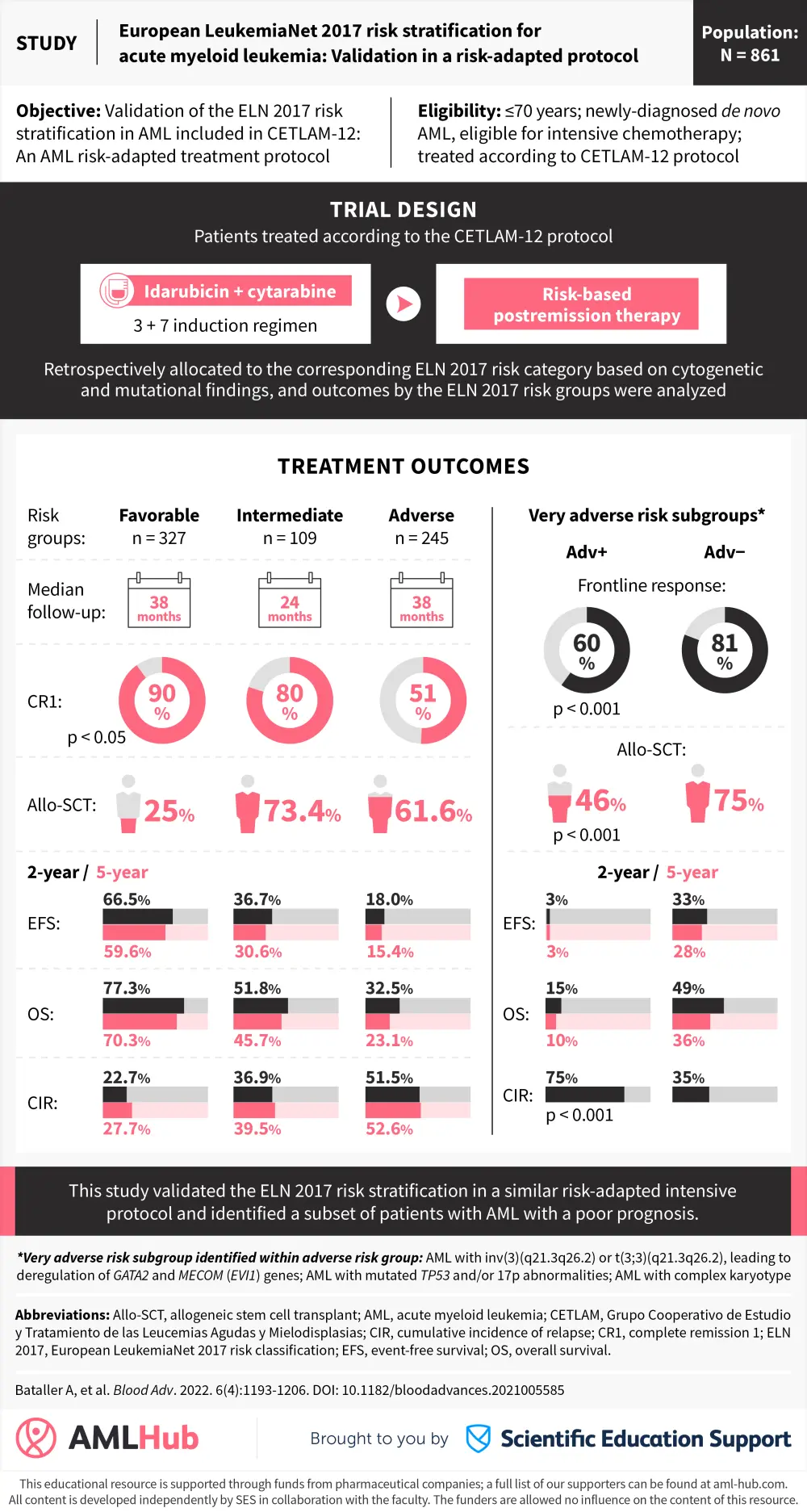
References
Please indicate your level of agreement with the following statements:
The content was clear and easy to understand
The content addressed the learning objectives
The content was relevant to my practice
I will change my clinical practice as a result of this content
Your opinion matters
Which AML-related topic do you currently need the most practical guidance on?

