All content on this site is intended for healthcare professionals only. By acknowledging this message and accessing the information on this website you are confirming that you are a healthcare professional. If you are a patient or carer, please visit Know AML.
The AML Hub website uses a third-party service provided by Google that dynamically translates web content. Translations are machine generated, so may not be an exact or complete translation, and the AML Hub cannot guarantee the accuracy of translated content. The AML Hub and its employees will not be liable for any direct, indirect, or consequential damages (even if foreseeable) resulting from use of the Google Translate feature. For further support with Google Translate, visit Google Translate Help.
The AML Hub is an independent medical education platform, sponsored by Daiichi Sankyo, Johnson & Johnson, Syndax, Thermo Fisher Scientific, Kura Oncology, and AbbVie. Funders are allowed no direct influence on our content. The levels of sponsorship listed are reflective of the amount of funding given. View funders.
Now you can support HCPs in making informed decisions for their patients
Your contribution helps us continuously deliver expertly curated content to HCPs worldwide. You will also have the opportunity to make a content suggestion for consideration and receive updates on the impact contributions are making to our content.
Find out more
Create an account to access:
Bookmark & personalize site content
Receive alerts for new content in your areas of interest
View AML content recommended for you
The effect of CD34+ cell dose on responses to haploidentical allo-HSCT: Results from a retrospective analysis
The graft cell dose following allogenic hematopoietic stem cell transplantation (allo-HSCT) has been suggested to influence patient outcome. Achievement of significant marrow engraftment following allo-HSCT is believed to rely heavily on the absolute number of infused donor cells, but the reliability of this statement remains unclear. Existing studies evaluating the impact of graft cell dose on different transplant outcomes have reached contrasting conclusions. Additionally, previous studies have concentrated on allo-HSCT from sibling and unrelated donors, whereas the influence of graft cell dose on outcomes with HSCT from haploidentical donors is less well investigated.
Enrico Maffini and colleagues set out to determine whether the dose of CD34+ cells in peripheral blood stem cell (PBSC) grafts correlates with resulting clinical outcomes following T-cell replete haploidentical allo-HSCT. To address this, they carried out a multicenter, retrospective analysis of patients receiving transplant for the treatment of acute myeloid leukemia (AML) during first and second complete remission (CR1/2) and found that a high graft cell dose may result in favorable outcomes in this patient group.
Study design
- Data from over 600 transplant centers were obtained from the European Society for Blood and Marrow Transplantation (EBMT) registry for retrospective analysis
- Adult patients (N = 414) receiving T-cell replete allo-HSCT from a haploidentical donor (> 2 human leukocyte antigen [HLA] mismatches) for the treatment of AML were eligible
- Patients received allo-HSCT between 2006–2018 and were in CR1/2
- Patients receiving ex vivo allo-HSCT were excluded from analysis
Study endpoints
- Neutrophil engraftment and platelet recovery
- Acute/chronic graft-versus-host disease (aGvHD/cGvHD)
- Patient survival outcomes: Relapse incidence (RI), non-relapse mortality (NRM), leukemia-free survival (LFS), overall survival (OS), and GvHD-free, relapse-free survival (GRFS)
Results
- Median follow-up: 23.3 months (range, 12.1–41.8)
Patient characteristics
- The eligible patients (Table 1) were divided into two cohorts depending on the CD34+ cell dose received
- High dose: > 4.96 × 106 CD34+ cells/kg (n = 334)
- Low dose: < 4.96 × 106 CD34+ cells/kg (n = 80)
Table 1. Baseline patient characteristics1
|
allo-HSCT, allogenic hematopoietic stem cell transplantation; ATG, anti-thymocyte globulin; CR1, first complete response; CR2, second complete response; MAC, myeloablative conditioning; PTCy, posttransplant cyclophosphamide; RIC, reduced intensity conditioning *At the time of allo-HSCT |
|
|
Characteristic |
Total patient cohort (N = 414) |
|---|---|
|
Median age, years (range) |
54 (18–74) |
|
Status at allo-HSCT, % CR1 CR2 |
70 30 |
|
sAML, % |
18 |
|
Cytogenetics, % Good Intermediate Unfavorable |
7 59 21 |
|
Karnofsky performance status ≥ 90*, % |
77 |
|
GvHD prophylaxis, % PTCy based ATG based |
71 29 |
|
Conditioning regimen, % MAC RIC |
43 57 |
Engraftment
- Median PBSC dose, per kg
- CD34+: 6.58 × 106 (interquartile range [IQR], 5.10–8.02)
- CD3+:2.60 × 108 (IQR, 1.84–3.63)
- Thirteen patients experienced graft failure
- Median time to platelet engraftment: 21 days (range, 1–37)
- GvHD prophylaxis regimen and CD34+ cell dose had a significant impact on both neutrophil and platelet engraftment
- Univariate analysis uncovered that patients receiving ATG as GvHD prophylaxis demonstrated superior engraftment to patients receiving PTCy (Figure 1A, B)
- Median time to neutrophil engraftment was significantly lower in patients receiving high dose CD34+ cells vs low dose (Figure 1C)
- Patients receiving an upper quartile CD3+ cell dose demonstrated lower rates of neutrophil (p = 0.025) and platelet (p = 0.031) engraftment compared to lower doses
Figure 1. Hematologic responses following allogenic hematopoietic stem cell transplantation1
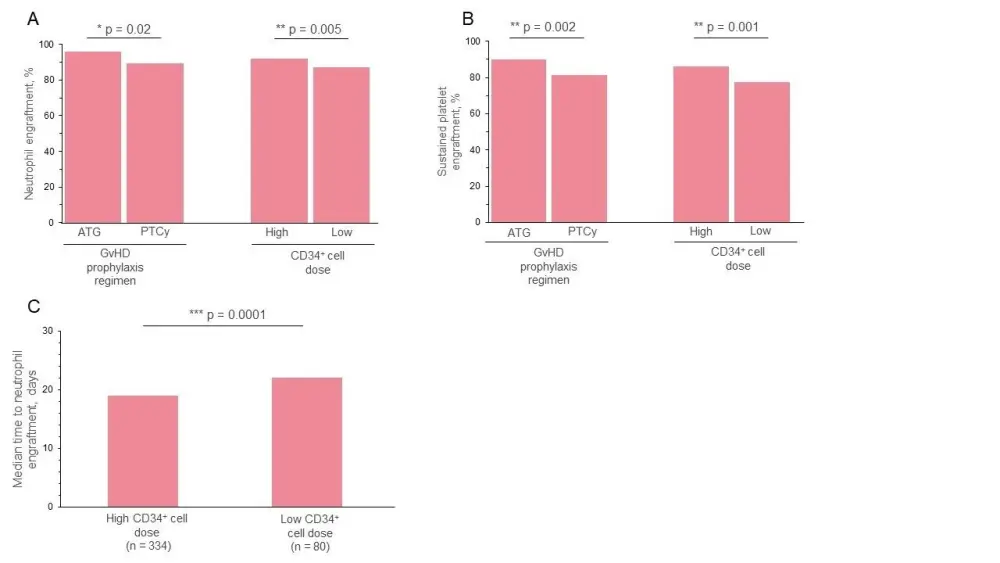
The impact of GvHD prophylaxis regimen and CD34+ cell dose on A neutrophil engraftment and B sustained platelet engraftment. C The influence of CD34+ cell dose on the time taken to reach neutrophil engraftment. Neutrophil engraftment was determined 30 days posttransplant. Sustained platelet engraftment was defined as > 20/uL at 6 months.
ATG, anti-thymocyte globulin; PTCy, posttransplant cyclophosphamide.
GvHD
- Incidences of aGvHD 100 days posttransplant:
- Grade 2–4: 32.3% (95% CI, 27.8–36.9)
- Grade 3–4: 14.6% (95% CI, 11.3–18.2)
- Incidences of two-year cGvHD:
- Overall: 36.3 % (95% CI, 30.9–41.6)
- Extensive: 14.4 % (95% CI, 10.7–18.6)
- Patients receiving ATG-based prophylaxis demonstrated a lower incidence Grade 2–4 aGvHD vs those receiving PTCy, (HR, 0.57; 95% CI, 0.37–0.88)
- Prophylaxis regimen had no influence over the incidence of Grade 3–4 GvHD
- A greater incidence of Grade 3–4 aGvHD was observed in the MAC vs RIC-receiving patient cohorts (18.9% vs 11.9%; p = 0.05)
- CD34+ dose had no effect on GvHD incidence
- Multivariant analysis uncovered that female donor to male recipient was a factor associated with increased incidence of extensive cGvHD when the cut-off point of 4.96 × 106 CD34+ cells was applied (HR, 2.21; 95% CI, 1.23–3.97; p = 0.008)
Patient outcomes
- Patient outcomes across the entire study cohort are presented in Table 2
- Patients receiving a high CD34+ cell dose demonstrated superior outcomes compared to the low dose cohort:
- NRM: HR, 0.48; 95% CI, 0.30–0.76
- LFS: HR, 0.63; 95% CI, 0.43–0.91
- OS: HR, 0.60; 95% CI, 0.40–0.88
- RI and GRFS were unaffected by the graft cell dose
- CD3+ and total nucleated cell (TNC) dose did not significantly impact posttransplant clinical outcomes
- Multivariate analysis uncovered additional prognostic factors for allo-HSCT
- Improved OS and NRM were observed in younger vs older patients*
- OS: 69.9% vs 59%; p = 0.043
- NRM: 18.9% vs 27.7%; p = 0.10
- Inferior survival and LFS were observed in patients receiving allo-HSCT in CR2 vs CR1
- Survival: HR, 1.46; 95% CI, 1.01–2.11; p = 0.0454
- LFS: HR, 1.39; 95% CI, 0.978–1.98; p = 0.0662
- Superior GRFS rates were observed in recipients of cytomegalovirus-positive transplant donors (HR, 0.75; 95% CI, 0.55–1.01; p = 0.058)
- Improved OS and NRM were observed in younger vs older patients*
*Stratified by 10-year age groups
Table 2. Two-year patient outcomes following allo-HSCT1
|
allo-HSCT, allogenic hematopoietic stem cell transplantation; GRFS, GvHD-free, relapse-free survival; LFS, leukemia-free survival; NRM, non-mortality relapse; OS, overall survival; RI, relapse incidence |
|
|
Patient response, % |
Total patient cohort (N = 414) |
|---|---|
|
OS |
64.5 |
|
LFS |
57.3 |
|
Disease RI |
19.5 |
|
NRM |
23.3 |
|
GRFS |
43.5 |
Conclusion
This analysis demonstrates that a CD34+ cell dose of > 4.96 × 106 cells/kg induces prolonged survival in patients with AML. The study suggests that the prolonged survival observed was a result of reduced NRM in these patients. However, when represented as a continuous variable, CD34+ cell dose had no impact on post-HSCT clinical outcome, and CD34+ cell levels above this had no further impact on survival, which concludes that the effect was non-linear. Although patients receiving high-dose CD34+ exhibited prolonged survival, the proportion of CD34+ cells appeared to have no impact on protection from disease recurrence, as RI rates remained the same.
The authors note the retrospective nature of the study as a substantial limitation of the analysis.
Nonetheless, the results from this study may help to refine the stem cell graft procedure from haploidentical donors to optimize patient survival outcomes.
References
Please indicate your level of agreement with the following statements:
The content was clear and easy to understand
The content addressed the learning objectives
The content was relevant to my practice
I will change my clinical practice as a result of this content
Your opinion matters
Which AML-related topic do you currently need the most practical guidance on?

