All content on this site is intended for healthcare professionals only. By acknowledging this message and accessing the information on this website you are confirming that you are a healthcare professional. If you are a patient or carer, please visit Know AML.
The AML Hub website uses a third-party service provided by Google that dynamically translates web content. Translations are machine generated, so may not be an exact or complete translation, and the AML Hub cannot guarantee the accuracy of translated content. The AML Hub and its employees will not be liable for any direct, indirect, or consequential damages (even if foreseeable) resulting from use of the Google Translate feature. For further support with Google Translate, visit Google Translate Help.
The AML Hub is an independent medical education platform, sponsored by Daiichi Sankyo, Johnson & Johnson, Syndax, Thermo Fisher Scientific, Kura Oncology, and AbbVie. Funders are allowed no direct influence on our content. The levels of sponsorship listed are reflective of the amount of funding given. View funders.
Now you can support HCPs in making informed decisions for their patients
Your contribution helps us continuously deliver expertly curated content to HCPs worldwide. You will also have the opportunity to make a content suggestion for consideration and receive updates on the impact contributions are making to our content.
Find out more
Create an account to access:
Bookmark & personalize site content
Receive alerts for new content in your areas of interest
View AML content recommended for you
Patterns of response to venetoclax + hypomethylating agents in IDH-mutated AML
Isocitrate dehydrogenase 1/2 (IDH1/2) mutations occur in around 20% of patients with acute myeloid leukemia (AML) and, as recently reported on the AML Hub, venetoclax (VEN) in combination with a hypomethylating agent (HMA), such as decitabine or azacitidine, is a promising therapeutic option for IDH-mutated AML.
Here, we are pleased to summarize a study by Danielle Hammond and colleagues, presented during the 62nd American Society of Hematology (ASH) Annual Meeting and Exposition.1 In this retrospective analysis, the patterns of response to VEN + HMA, including measurable residual disease (MRD) by flow cytometry (FC) and next generation sequencing (NGS), were evaluated in patients with IDH-mutated AML.
Study design
A single-institution, retrospective analysis of 65 patients with newly diagnosed or relapsed/refractory (R/R) IDH1/2-mutated AML who had been treated with VEN in combination with HMA.
Primary analyses:
- Responses according to modified 2017 European LeukemiaNet criteria, incorporating MRD assessment by FC (sensitivity 10-3–10-4).
- NGS MRD by mutant IDH variant allele frequency assessment of bone marrow samples pre-treatment and at time of best response.
Secondary analysis:
- Comparative response in a subset of patients treated with IDH inhibitor (IDHi)-based regimens.
Key findings
Selected patient, disease, and treatment characteristics are shown in Table 1.
- Almost three-quarters of patients had an IDH2 mutation; commonly co-mutated genes included SRSF2, RAS-pathway, and NPM1.
- Decitabine was the HMA administered in 85% of cases.
- Common reasons for VEN + HMA discontinuation (n = 57) were relapse (30%), resistant/inadequate response (18%), stem cell transplant (25%), and patient/physician choice (20%).
- Approximately half of patients had received previous therapies; 15% had received prior IDHi treatment, and 40% had received treatment with IDHi following VEN + HMA.
Table 1. Selected patient, disease, and treatment characteristics1
|
AML, acute myeloid leukemia; del, deletion; ELN, European LeukemiaNet; FLT3, FMS-like tyrosine kinase 3; HMA, hypomethylating agent; IDH, isocitrate dehydrogenase; IDHi, IDH inhibitor; ITD, internal tandem duplication; NPM1, nucleophosmin-1; RAS, rat sarcoma; VEN, venetoclax. |
|
|
Characteristic |
N = 65 |
|---|---|
|
Median age, years (range) |
69 (32–83) |
|
2017 ELN prognostic group, % |
|
|
Cytogenetics, % |
|
|
IDH mutational status, % |
|
|
Co-mutations, % |
|
|
Prior therapies for AML, % |
|
|
Median number of VEN + HMA cycles (range) |
3 (1–34) |
|
IDHi therapy |
|
Response rates, MRD, and overall survival
Best bone marrow response rates after a median of one treatment cycle (range, 1–5) are shown in Figure 1.
Figure 1. Best response rates1
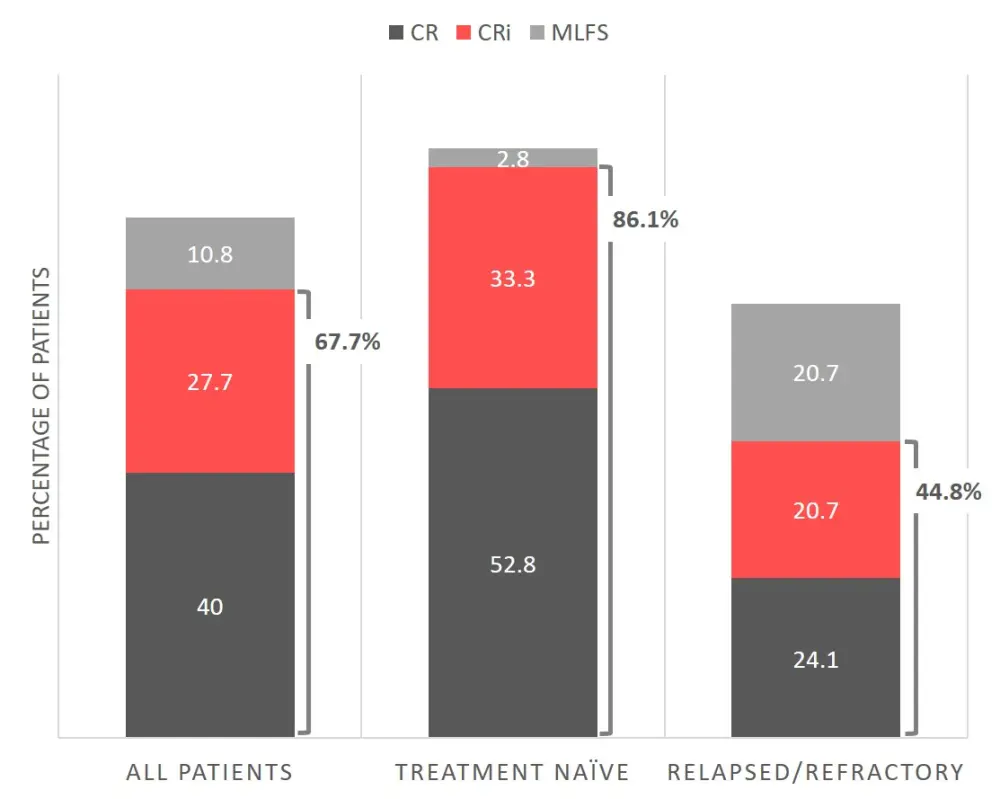
CR, complete response; CRi, CR with incomplete hematologic recovery; MLFS, morphologic leukemia-free state.
Overall, 67.7% of patients achieved a complete response (CR) or CR with incomplete hematologic recovery (CRi) after VEN + HMA treatment. Of all patients achieving CR/CRi, 90% were also MRD-negative by FC, whereas only 59% were MRD-negative by NGS. Median duration of response was 18.0 months (95% CI, 13.7–NE).
Responses were deeper and more durable for patients receiving frontline VEN + HMA therapy compared with the R/R setting (CR/CRi 86.1% vs 44.8%, and median duration of response 24.1 months vs 15.1 months). After frontline therapy, 90% of CR/CRi responses were FC MRD-negative, compared with 42% MRD negativity by NGS. MRD responses were less robust in the R/R setting, with just 62.5% and 50% of CR/CRi responses also negative by FC and NGS MRD, respectively.
Median overall survival (OS) for all patients treated with VEN + HMA was 22.4 months (95% CI, 16.4–NE), and was not reached (95% CI, 42.1–NE) for patients who had received frontline VEN + HMA. There was a significant association between improved OS and depth of response by bone marrow assessment (p < 0.001) and MRD negativity by FC (p < 0.001) (Table 2). In contrast, MRD negativity by mutant IDH NGS was not associated with a survival benefit (p = 0.385), however mutant IDH variant allele frequency data was only available in 60% of responding patients and analysis of additional samples is underway.
Table 2. Overall survival by depth of response1
|
|
Median OS, months (95% CI) |
|---|---|
|
CR, complete response; CRi, CR with incomplete hematologic recovery; FC, flow cytometry; mIDH, mutant isocitrate dehydrogenase; MLFS, morphologic leukemia-free state; MRD, measurable residual disease; NGS, next generation sequencing; NE, not evaluable; NR, not reached; OS, overall survival. |
|
|
All patients |
22.4 (16.4–NE) |
|
By marrow assessment |
|
|
CR |
NR (NE–NE) |
|
CRi |
42.2 (20.1–NE) |
|
MLFS |
13.4 (7.9–NE) |
|
No response |
5.3 (3.5–NE) |
|
By FC MRD assessment* |
|
|
FC MRD-negative |
NR (42.2–NE) |
|
FC MRD-positive |
15.7 (2.8–NE) |
|
By mIDH NGS MRD assessment* |
|
|
mIDH NGS MRD-negative |
NR (NE–NE) |
|
mIDH NGS MRD-positive |
NR (20.2–NE) |
Response by co-mutation pattern
NPM1 mutations were enriched in patients with superior bone marrow responses and MRD-negative status. Conversely, RAS-pathway and TP53 mutations were enriched in patients with poorer responses and MRD-positive status.
Impact of IDHi treatment
Of 29 patients treated with an IDHi-based regimen, eight patients were treated with IDHi initially, and then switched to VEN + HMA therapy, mainly due to lack or loss of response, and 21 received IDHi as a salvage regimen after VEN + HMA.
A bone marrow response was seen in seven of the eight patients (88%) who received VEN + HMA as salvage therapy, and in 11/21 (52%) patients who received IDHi as salvage therapy after VEN + HMA; the highest response in this setting was seen when an IDHi was added to the VEN + HMA backbone.
Conclusion
VEN + HMA is an effective treatment option for IDH-mutated AML, particularly for patients with co-mutated NPM1 and in the frontline setting, where CR/CRi rates reached 86.1%. MRD negativity by FC was frequent and predictive of superior OS, however, IDH1/IDH2 mutations by NGS were still detectable in around half of patients at best response, highlighting a potential role for combination therapy with an IDHi. Further research into combination vs sequential therapy with VEN + HMA and IDHi-based regimens is warranted.
References
Please indicate your level of agreement with the following statements:
The content was clear and easy to understand
The content addressed the learning objectives
The content was relevant to my practice
I will change my clinical practice as a result of this content
Your opinion matters
Which AML-related topic do you currently need the most practical guidance on?

