All content on this site is intended for healthcare professionals only. By acknowledging this message and accessing the information on this website you are confirming that you are a healthcare professional. If you are a patient or carer, please visit Know AML.
The AML Hub website uses a third-party service provided by Google that dynamically translates web content. Translations are machine generated, so may not be an exact or complete translation, and the AML Hub cannot guarantee the accuracy of translated content. The AML Hub and its employees will not be liable for any direct, indirect, or consequential damages (even if foreseeable) resulting from use of the Google Translate feature. For further support with Google Translate, visit Google Translate Help.
The AML Hub is an independent medical education platform, sponsored by Daiichi Sankyo, Johnson & Johnson, Syndax, Thermo Fisher Scientific, Kura Oncology, and AbbVie. Funders are allowed no direct influence on our content. The levels of sponsorship listed are reflective of the amount of funding given. View funders.
Now you can support HCPs in making informed decisions for their patients
Your contribution helps us continuously deliver expertly curated content to HCPs worldwide. You will also have the opportunity to make a content suggestion for consideration and receive updates on the impact contributions are making to our content.
Find out more
Create an account and access these new features:
Bookmark content to read later
Select your specific areas of interest
View AML content recommended for you
Patients with poor risk cytogenetics and TP53wt treated with venetoclax and HMA show increases in overall survival compared to treatment with azacitidine only
Patients with acute myeloid leukemia (AML) with poor-risk cytogenetics show sub optimal responses to conventional therapies. Patients carrying a TP53 mutation are highly correlated with poor risk cytogenetic profiles and is independently associated with adverse outcomes when treated with venetoclax. The treatment option of venetoclax plus azacitidine is the standard regimen for patients who are unsuitable for intensive induction chemotherapy. However, the outcome of using hypomethylating agents (HMA) and venetoclax to treat patients with AML who have poor-risk cytogenetics and who harbour either TP53mut or TP53wt, is unknown.
During the 63rd American Society of Hematology (ASH) Annual Meeting and Exposition, Daniel Pollyea, University of Colorado School of Medicine, Aurora, US, presented results from a study where the safety and efficacy of venetoclax plus azacitidine or HMA were assessed in patients with treatment-naïve AML, who had poor-risk cytogenetics and who harboured either TP53mut or TP53wt.1 We summarize key results below.
Study design1
Treatment-naïve patients with AML who were not suitable for chemotherapy due to co-morbidities or being ≥75 years, were enrolled.
Data were combined from the ongoing phase III VIALE-A trial (NCT02993523) comparing venetoclax + azacitidine and placebo + azacitidine, and a phase Ib study (NCT02203773) where patients were treated with venetoclax + HMA (azacitidine or decitabine). Treatment was organized into a 28-day cycle; 400 mg venetoclax was administered daily, 75 mg/m2 azacitidine on Days 1–7, and 20 mg/m2 decitabine on Days 1–5.
Cytogenetics risk was determined pre-treatment as per the National Comprehensive Cancer Network (NCCN) criteria, with several abnormalities found to be associated with poorer outcomes. These included three or more clonal chromosomal abnormalities, monosomal karyotype, and chromosomal band 11q23 translocation. TP53 status was also assessed at screening and analyzed using MyAML panel assays. Patients with inconclusive or missing results were excluded.
Key endpoints were complete remission (CR) + CR with incomplete hematologic remission (CRi), duration of remission (DoR), and overall survival (OS).
Results
The pooled data included 546 patients, of which 353 were treated with venetoclax + azacytidine, and 145 with azacitidine only. A total of 127 patients in the venetoclax + azacitidine treatment group were found to have poor cytogenetics versus 56 patients in the azacitidine treatment group. A further 401 patients were treated with venetoclax + HMA; of these, 152 were found to have poor cytogenetics. The median variant allele frequency for TP53 was 42.9% for both venetoclax + azacitidine and azacitidine alone.
In patients with poor-risk cytogenetics and TP53wt, the composite complete remission (CRc) rate in the venetoclax + azacitidine group was 70% compared to 23% in the azacitidine group. The median DoR was 18.37 months for patients treated with venetoclax + azacitidine, and 8.51 months for those treated with azacitidine alone. The median OS for the venetoclax + azacitidine treatment group was 23.43 months compared to 11.29 months in the azacitidine group. Results were similar for patients treated with venetoclax + HMA; the CRc rate was found to be 74%, the median DoR was 16.72 months, and the median OS was 21.13 months. These results were also comparable to those patients with intermediate-risk cytogenetics and TP53wt.
For patients with poor-risk cytogenetics and TP53mut, the CRc rate in the venetoclax + azacitidine group was 41% versus 17% for patients in the azacitidine group. The median DoR was 6.54 months for patients treated with venetoclax + azacytidine, and 6.7 months for those treated with azacitidine alone. The median OS was 5.17 months for patients in the venetoclax + azacitidine group compared to 4.9 months in the azacitidine group. Patients treated with venetoclax + HMA were found to have a CRc rate of 47%, and a median DoR and OS of 6.54 and 5.42 months, respectively. CRc rate for each group can be seen in Figure 1, and median DoR and OS are shown in Figure 2.
Figure 1. CRc rates in patients with either TP53wt or TP53mut per treatment group*
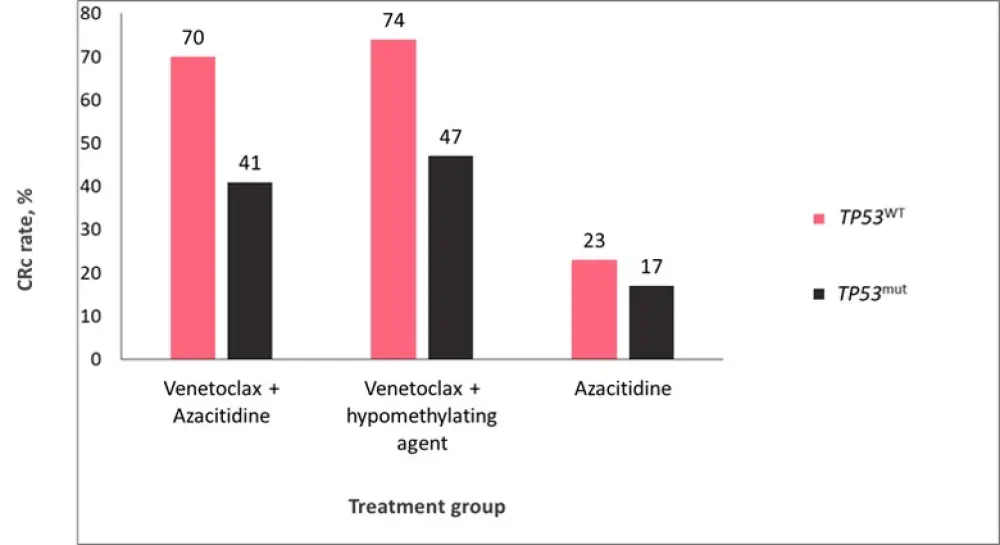
CRc, composite complete remission.
*Adapted from Pollyea et al.1
Figure 2. Median DoR and OS in patients with either TP53wt or TP53mut per treatment group*
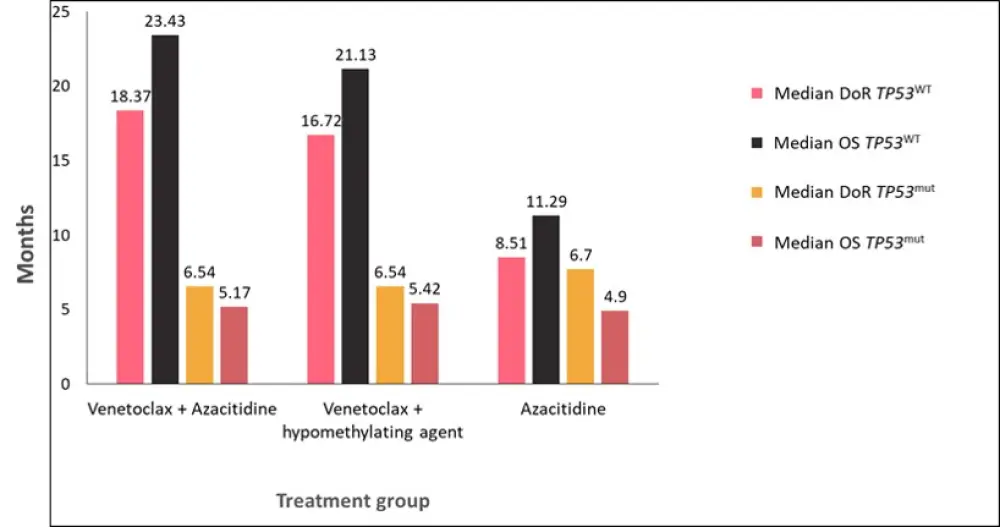
DoR, duration of remission; OS, overall survival.
*Adapted from Pollyea et al.1
Common Grade 3 adverse events (AEs) in patients with poor-risk cytogenetics and TP53wt, treated with either venetoclax + azacitidine or azacitidine alone, included thrombocytopenia (36% in both groups), febrile neutropenia (32% and 14%, respectively), and pneumonia (18% and 23%, respectively). Patients with TP53mut also experienced thrombocytopenia (28% for both groups), febrile neutropenia (43% and 22%, respectively), and pneumonia (26% and 53%, respectively). Patients treated with Ven + HMA also reported similar Grade 3 events.
Conclusion
Patients with poor-risk cytogenetics and TP53wt treated with either venetoclax + azacitidine or venetoclax + HMA showed notable increases in remission rates, DoR, and OS compared with azacitidine only. In contrast, patients with poor-cytogenetic risk and TP53mut did not show significant improvement in DoR or OS in any treatment group. No new toxicities were reported.
References
Please indicate your level of agreement with the following statements:
The content was clear and easy to understand
The content addressed the learning objectives
The content was relevant to my practice
I will change my clinical practice as a result of this content

