All content on this site is intended for healthcare professionals only. By acknowledging this message and accessing the information on this website you are confirming that you are a healthcare professional. If you are a patient or carer, please visit Know AML.
The AML Hub website uses a third-party service provided by Google that dynamically translates web content. Translations are machine generated, so may not be an exact or complete translation, and the AML Hub cannot guarantee the accuracy of translated content. The AML Hub and its employees will not be liable for any direct, indirect, or consequential damages (even if foreseeable) resulting from use of the Google Translate feature. For further support with Google Translate, visit Google Translate Help.
The AML Hub is an independent medical education platform, sponsored by Daiichi Sankyo, Johnson & Johnson, Syndax, Thermo Fisher Scientific, Kura Oncology, and AbbVie. Funders are allowed no direct influence on our content. The levels of sponsorship listed are reflective of the amount of funding given. View funders.
Now you can support HCPs in making informed decisions for their patients
Your contribution helps us continuously deliver expertly curated content to HCPs worldwide. You will also have the opportunity to make a content suggestion for consideration and receive updates on the impact contributions are making to our content.
Find out more
Create an account and access these new features:
Bookmark content to read later
Select your specific areas of interest
View AML content recommended for you
Outcomes with venetoclax in treating AML in real-world settings
The combination of venetoclax and hypomethylating agents has significantly improved outcomes and survival in patients with newly diagnosed acute myeloid leukemia (AML) who are ineligible for intensive chemotherapy.1 There is a continuous effort to investigate venetoclax further in alternative settings (e.g., combinations with other conventional agents), and in patients with poor risk cytogenetics, and relapsed/refractory (R/R) disease. The AML Hub has been providing an extensive coverage on the venetoclax trials, which can be found here.
Within our recent educational theme on venetoclax combinations in the AML setting, we summarize safety and efficacy findings of venetoclax from a retrospective analysis carried out in newly diagnosed or previously treated patients at an academic site; the results were published by Fleischmann et al.1 in the Journal of Cancer Research and Clinical Oncology.
Study design
A total of 56 consecutive patients (Table 1) were given venetoclax in a 28-day cycle in combination with:
- Hypomethylating agents (HMAs): Decitabine 20 mg/m2/day intravenously on Days 1–5 or azacitidine 75 mg/m2 subcutaneously for 7 days, OR
- Low-dose cytarabine (LDAC) 40 mg subcutaneously on Days 1–7
Conditioning regimens before allogeneic hematopoietic stem cell transplantation (allo-HSCT) included a reduced-toxicity conditioning (81.8% of patients) and myeloablative conditioning (18.2% of patients).
Venetoclax was given in different treatment cohorts and disease settings (Figure 1). Overall survival (OS), relapse-free survival (RFS), and overall response rate (ORR) of composite complete remission (CRc) + partial remission, were evaluated.
Figure 1. Venetoclax treatment diagram*
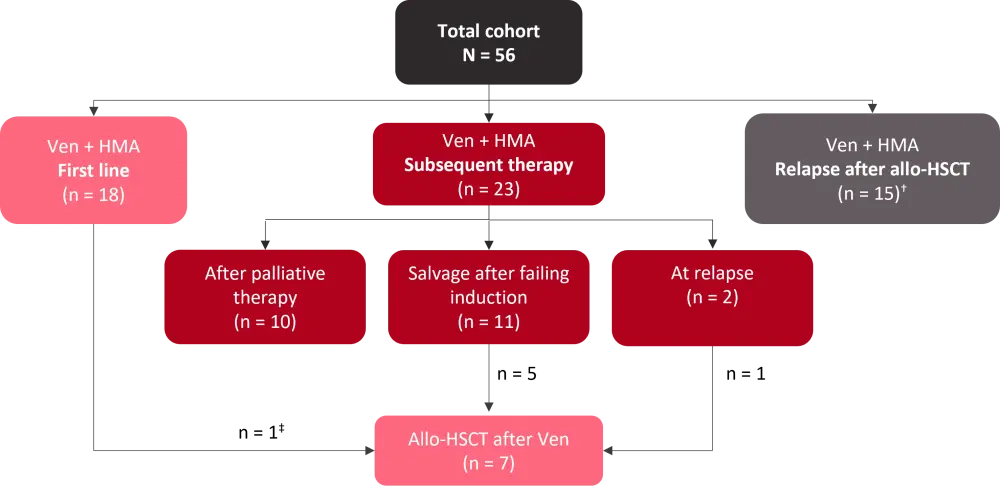
Allo-HSCT, allogeneic hematopoietic stem cell transplantation; HMA, hypomethylating agent; Ven, venetoclax.
*Adapted from Fleischmann et al.1
†Includes two cases at relapse after second allo-HSCT.
‡Received Ven-HMA with intention to allo-HSCT.
Table 1. Patient characteristics*
|
AML, acute myeloid leukemia; ECOG PS, Eastern Cooperative Oncology Group performance status; ELN, European LeukemiaNet; s-AML, secondary AML; t-AML, therapy-related AML. |
|
|
Characteristic, % unless otherwise stated |
N = 56 |
|---|---|
|
Median age, years (range) |
66.5 (34–83) |
|
Female |
42.8 |
|
ECOG PS |
|
|
0–1 |
55.4 |
|
2–3 |
44.6 |
|
AML diagnosis |
|
|
de novo |
48.2 |
|
s-AML |
51.8 |
|
t-AML |
1.8 |
|
Cytogenetic risk† |
|
|
Favorable |
0 |
|
Intermediate |
60.7 |
|
Adverse |
37.5 |
|
Unknown |
1.8 |
|
ELN risk category |
|
|
Favorable |
8.9 |
|
Intermediate |
30.3 |
|
Adverse |
57.1 |
|
Not available |
3.6 |
Next-generation sequencing (NGS), performed in 54 patients, showed that at the initiation of venetoclax, 33.9% had no aberrations, followed by 16% with NPM1 mutation, 14.3% with FLT3-ITD mutations, and 12.5% with IDH1/2 mutations. TP53 mutations were detected in 8.9% of patients.
Results
Median follow-up was 11.5 months (range, 6.1 – 22.3):
- Median OS at the initiation of venetoclax treatment across cohorts
- First line: 13.3 months (2.2–20.5)
- Subsequent line: 5.0 months (0.8–24.3)
- At relapse post-allo-HSCT: 4.0 months (1.5–22.1)
- Prior to allo-HSCT: 11.5 months (10.4–22.3)
- Median OS and progression-free survival (PFS) in patients achieving CRc were 20.8 months (range, 2.5–24.3) and 20.5 months (range, 2.6–24.3), respectively.
- Median RFS was 10.2 months (2.2–24.3)
- ORR for the whole cohort was 51.8% with a median time to first response in patients achieving CRc of 2.7 months (1.4–16.2).
- First-line: 61.1%
- Subsequent line: 52.2%
- At relapse post-allo-HSCT: 42.8%
- There was a decrease in transfusion of platelets and red blood cells required at Day 100 from venetoclax initiation.
- Platelet: 63% → 47%
- RBC: 76% → 55%
Analysis in different genetic subgroups
- ORR in patients with intermediate-risk and high-risk cytogenetics were 57.5% and 42.8%, respectively.
- FLT3-ITD mutation: 12.5%
- NPM1 and/or IDH1/2 mutation: 71.4%
- There was no difference in median OS among cytogenetic risk groups and ELN risk groups.
- FLT3-ITD mutation was associated with a significantly inferior median OS of 3.4 months (vs 10.4 months in case of no FLT3-ITD mutation, p = 0.0002).
- Median OS in patients with NPM1, IDH1/2 mutations without a FLT3-ITD mutation was 11.2 months (vs 5.0 months in the remaining cohort; p = 0.131), indicating an increased sensitivity to venetoclax therapy.
Outcomes by venetoclax dosage
The overall mean venetoclax dose was 149 mg (20.9–362.5) and the median treatment duration was 105 days (range, 18–674). Outcomes from patients with a mean daily dose of ≤100 mg were compared to those receiving >100 mg to evaluate the impact of venetoclax dosage (Table 2).
Table 2. Comparison of outcomes by venetoclax dose groups*
|
ORR, overall response rate; OS, overall survival; PD, progressive disease. |
||
|
Outcomes |
Patients receiving ≤100 mg |
Patients receiving >100 mg |
|---|---|---|
|
Median OS, months |
6.4 |
8.1 |
|
ORR, % |
45.5 |
55.9 |
|
PD on venetoclax, % |
22.3 |
8.9 |
Safety
Grade 3–4 adverse events (AEs) included:
- Neutropenia: Grade 3 = 15%; Grade 4 = 30%
- Thrombocytopenia: Grade 3 = 15%
- Anemia: Grade 3 = 12%
- Renal insufficiency: Grade 3 = 5%
- Fatigue: Grade 3 = 7%
- Nausea: Grade 3 = 9%
Nonrelapse mortality rate was 8.9% (n = 5) and two patients died in the first 30 days of treatment.
Conclusion
In this study, investigators attempted to clarify the efficacy of venetoclax-based combinations in real-world settings, commonly encountered in clinical practice. The findings demonstrated that the venetoclax-based combination approach was associated with encouraging response rates in first-line settings. Response rates were also found to be promising in relapsed/refractory AML, including relapse after allo-HSCT. Salvage treatment with venetoclax may allow patients failing conventional chemotherapy to have a potential curative allo-HSCT.
References
Please indicate your level of agreement with the following statements:
The content was clear and easy to understand
The content addressed the learning objectives
The content was relevant to my practice
I will change my clinical practice as a result of this content
Your opinion matters
Which AML-related topic do you currently need the most practical guidance on?

