All content on this site is intended for healthcare professionals only. By acknowledging this message and accessing the information on this website you are confirming that you are a healthcare professional. If you are a patient or carer, please visit Know AML.
The AML Hub website uses a third-party service provided by Google that dynamically translates web content. Translations are machine generated, so may not be an exact or complete translation, and the AML Hub cannot guarantee the accuracy of translated content. The AML Hub and its employees will not be liable for any direct, indirect, or consequential damages (even if foreseeable) resulting from use of the Google Translate feature. For further support with Google Translate, visit Google Translate Help.
The AML Hub is an independent medical education platform, sponsored by Daiichi Sankyo, Johnson & Johnson, Syndax, Thermo Fisher Scientific, Kura Oncology, and AbbVie. Funders are allowed no direct influence on our content. The levels of sponsorship listed are reflective of the amount of funding given. View funders.
Now you can support HCPs in making informed decisions for their patients
Your contribution helps us continuously deliver expertly curated content to HCPs worldwide. You will also have the opportunity to make a content suggestion for consideration and receive updates on the impact contributions are making to our content.
Find out more
Create an account to access:
Bookmark & personalize site content
Receive alerts for new content in your areas of interest
View AML content recommended for you
Low intensity decitabine plus venetoclax versus IC in patients with R/R AML: A propensity score matched analysis
Patients with relapsed or refractory (R/R) acute myeloid leukemia (AML) have no standard treatments and have dismal outcomes with 10–20% long-term survival. Continuing intensive chemotherapy (IC) in these patients becomes challenging due to disease progression or subsequent therapy, including stem cell transplantation (SCT). Thereby, novel, lower intensity chemotherapies are warranted. Venetoclax (VEN)-based lower intensity regimens are standard treatments for older or unfit patients with AML and are potentially efficacious in younger patients as well. However, there is no evidence of how venetoclax-based lower intensity regimens compare with IC.
The AML Hub has previously reported an expert interview describing the real-word experience of R/R AML treatment with venetoclax plus hypomethylating agents (HMAs) and an article on the effectiveness of decitabine (DEC) + VEN in patients with high-risk AML and myelodysplastic syndromes (MDS). Here we present the key findings of a recent study by Maiti et al.1, published in Cancer, comparing the outcomes of low-intensity DEC + VEN versus IC in patients with R/R AML.
Study design
This was a retrospective, cohort study consisting of two cohorts, the 10-day decitabine and venetoclax (DEC10-VEN) cohort from the prospective phase II trial of DEC10-VEN (NCT03404193), and the IC cohort. Patients enrolled in both the DEC10-VEN and IC cohorts were adults aged >18 years with an Eastern Cooperative Oncology Group performance status (ECOG PS) of ≤ 3.
Treatment regimens:
- Patients in the DEC10-VEN were treated between 2018 and 2020 and received 20 mg/m2 decitabine daily for 10 days every 4–6 weeks with 40 mg venetoclax daily as an induction therapy.
- Patients received decitabine for 5 days and venetoclax daily for consolidation once they achieved a response.
- Patients in the IC cohort were treated between 2005 and 2019 and received salvage therapy with idarubicin with cytarabine (IA), with or without cladribine (CLIA), clofarabine (CIA), or fludarabine (FIA or FLAG-IDA)-based regimens without venetoclax.
The included IC regimens comprised of the following therapies given intravenously:
- IA-based regimen: 10–12 mg/m2 idarubicin on Days 1 to 3, and 1–2 g/m2 cytarabine on Days 1 to 4.
- CLIA-based regimen: 5 mg/m2 of cladribine on Days 1 to 5, 1–2 g/m2 cytarabine on Days 1–5, and 10 mg/m2 idarubicin on Days 1–3.
- CIA-based regimen: 15 mg/m2 clofarabine daily on Days 1–5, 10 mg/m2 idarubicin daily on days 1–3, and 1 g/m2cytarabine daily on Days 1–5.
- FLAG-IDA or FIA-based regimens: 30 mg/m2 fludarabine on Days 1 to 5, 10 mg/m2 idarubicin daily on Days 1 to 3, and 1–2 g/m2 cytarabine daily on Days 1 to 5, with or without 5 µg/kg filgrastim subcutaneously on Days 1–7.
Outcomes included:
- Overall response rate (ORR) including complete remission (CR), CR with incomplete hematologic recovery (CRi), and morphologic leukemia-free state (MLFS).
- Minimal residual disease (MRD)
- Event-free survival (EFS): From start of therapy to the date of primary refractory disease, relapse from CR/CRi, or death from any cause.
- Overall survival (OS): From start of therapy until death or censorship at last follow-up.
- Relapse-free survival (RFS): From the date of achieving CR/CRi until relapse, death, or censorship at last follow-up.
- Adverse events (AEs)
Results
Baseline characteristics
The best matched patients using propensity scores between the two cohorts is shown in Figure 1.
Figure 1. Propensity score matching*
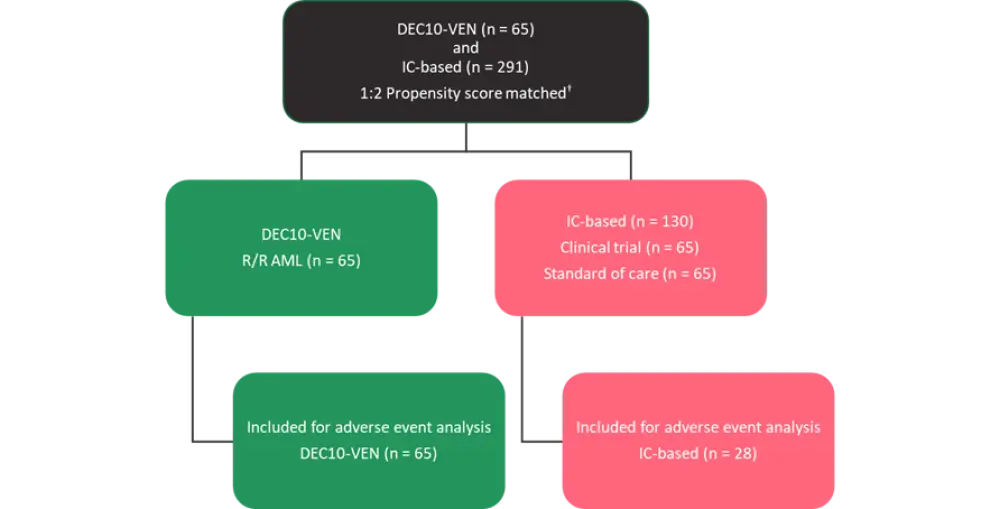
AML, acute myeloid leukemia; IC, intensive chemotherapy; DEC10-VEN, 10-day decitabine with venetoclax; R/R, relapsed or refractory.
*Adapted from Maiti et al.1
†Propensity score matched for age, Eastern Cooperative Oncology Group performance status (ECOG PS), number of therapies, AML (primary refractory vs relapsed), first complete remission (CR1) duration, prior stem cell transplantation (SCT), European LeukemiaNet cytogenetic risk category, FLT3-ITD/TKD (mutated vs wild type), TP53 (mutated vs wild type).
The median age was 64 years (range, 18–85) and 58 years (range, 19–80) in the DEC10-VEN and IC cohorts, respectively. Patients received a median of two cycles (range, 1–16) and one cycle (range, 1–5) of therapy in the DEC10-VEN and IC cohorts, respectively. The baseline characteristics were well balanced between the two cohorts (Table 1).
Table 1. Baseline characteristics*
|
AHD, antecedent hematologic disorder; AML, acute myeloid leukemia; DEC10-VEN, 10-day decitabine with venetoclax; ECOG PS, Eastern Cooperative Oncology Group performance status; ELN, European LeukemiaNet; HMA, hypomethylating agent; IC, intensive chemotherapy; allo-SCT, allogeneic stem cell transplantation. |
|||
|
Characteristic, % unless otherwise stated |
DEC10-VEN |
IC |
p value |
|---|---|---|---|
|
Age, <60 years |
40 |
57 |
0.038 |
|
Sex, male |
60 |
55 |
0.539 |
|
AML type |
|||
|
Primary refractory |
43 |
42 |
— |
|
Relapsed |
57 |
58 |
0.837 |
|
Secondary AML from AHD |
17 |
16 |
0.891 |
|
ECOG PS |
|||
|
0–1 |
71 |
73 |
0.734 |
|
≥2 |
29 |
27 |
— |
|
Median bone marrow blasts (range) |
34 (1–96) |
35 (0–98) |
0.536 |
|
ELN 2017 cytogenetic |
|||
|
Favorable |
0 |
0 |
— |
|
Intermediate |
63 |
62 |
0.958 |
|
Adverse |
37 |
38 |
— |
|
Mutations |
|||
|
NPM1 |
22 |
12 |
0.605 |
|
FLT3-ITD/TKD |
25 |
26 |
— |
|
IDH1/2 |
17 |
22 |
— |
|
TP53 |
28 |
25 |
— |
|
RUNX1 |
18 |
17† |
— |
|
ASXL1 |
15 |
11† |
— |
|
ELN 2017 risk group |
|||
|
Favorable |
15 |
8 |
0.302 |
|
Intermediate |
18 |
23 |
— |
|
Adverse |
66 |
68 |
— |
|
Median no. of prior therapies (range) |
2 (1–8) |
2 (1–10) |
0.721 |
|
Prior therapies |
|||
|
HMA |
25 |
18 |
0.214 |
|
IC only |
48 |
41 |
— |
|
IC and HMA |
25 |
41 |
— |
|
allo-SCT |
28 |
28 |
— |
Efficacy
- At a median follow-up of 17.5 and 49.3 months in the DEC10-VEN and IC cohorts, respectively, patients in the DEC10-VEN cohort had a higher ORR (60% vs 36%; p < 0.001) (Table 2).
- CRi rates, MLFS rates, and MRD negativity were also similar to ORR and higher in the DEC10-VEN cohort. In addition, the rate of refractory disease was significantly lower in the DEC10-VEN compared to IC cohort (Table 2).
- The median EFS was 5.7 and 1.5 months (HR, 0.46; p = < 0.001) and the median OS was 6.8 and 4.7 months (HR, 0.56; p = 0.008) in the DEC10-VEN and IC cohorts, respectively.
- The median OS was numerically higher in DEC10-VEN compared to the IC cohort (18.7 vs 12.9 months, HR, 1.11; p = 0.813).
- Exploratory subgroup analysis showed that DEC10-VEN was comparable with IC as salvage therapy for R/R AML.
Table 2. Efficacy outcomes*
|
CR, complete remission; CRi, CR with incomplete hematologic recovery; DEC10-VEN, 10-day decitabine with venetoclax; HR, hazard ratio; IC, intensive chemotherapy; MLFS, morphologic leukemia-free state; MRD, minimal residual disease; OR, odds ratio; ORR, CR + CRi + MLFS. |
||||
|
Outcomes |
DEC10-VEN |
IC (n = 130) |
OR/HR |
p value† |
|---|---|---|---|---|
|
ORR |
60 |
28 |
3.28 |
< 0.001 |
|
CR/CRi |
42 |
23 |
2.52 |
0.009 |
|
CR |
23 |
17 |
1.44 |
0.320 |
|
CRi |
19 |
6 |
3.56 |
0.012 |
|
MLFS |
15 |
5 |
3.33 |
0.020 |
|
MRD negativity |
28 |
13 |
2.48 |
0.017 |
|
Refractory |
35 |
55 |
0.46 |
0.011 |
|
Relapse |
51 |
47 |
0.75 |
0.630 |
|
30-day mortality |
8 |
12 |
0.58 |
0.322 |
|
60-day mortality |
12 |
27 |
0.40 |
0.029 |
- The median OS in patients with first complete remission (CR1) duration of >6 months was 21.8 and 6.5 months in the DEC10-VEN and IC cohorts, respectively (HR, 0.52; p = 0.023).
- Median OS was also higher (6.7 vs 3.5 months) in DEC10-VEN compared to the IC cohort in patients with antecedent hematologic disorder (HR, 0.42; p = 0.017).
- DEC10-VEN was independently associated with significantly superior OS (HR, 0.66; p = 0.012) in a multivariate analysis of an unmatched population, while IDH1/2 mutation was associated with a lower risk of death (HR, 0.70; p = 0.029).
- The risk of death was higher in patients with antecedent hematologic disorder (HR, 1.84; p < 0.001), European LeukemiaNet (ELN) adverse-risk cytogenetics (HR, 1.57; p < 0.001), TP53 mutation (HR, 1.71; p < 0.001), and failure of two or more lines of therapy.
Safety
- The 60-day mortality was 12% vs 27% in the DEC10-VEN and IC cohorts, respectively (odds ratio [OR], 0.40; p = 0.029) (Table 2).
- Nonhematologic Grade 3 and 4 treatment-emergent AEs occurred in 21 (75%) of 28 patients receiving IC and 45 (69%) of 65 patients receiving DEC10-VEN (p = 0.628).
- Grade 3–4 infectious AEs occurred in 25 out of 65 (38%) and 5 out of 28 (18%) patients in the DEC10-VEN and IC cohorts, respectively.
- Febrile neutropenia occurred in 30% vs 50% of patients in the DEC10-VEN and IC cohorts, respectively (p = 0.101).
Conclusion
This retrospective cohort study demonstrated that DEC10-VEN can achieve high ORR, rates of CR, CRi, MLFS, MRD negativity, and better OS, and EFS. DEC10-VEN, being a low-intensity regimen, has the potential to act as a bridge to SCT. Taken the findings together, DEC10-VEN may be an appropriate salvage option for venetoclax-naïve patients with R/R AML. Although, propensity score matching minimized any biases, the study was still limited by a small number of patients in the DEC10-VEN cohort for any exploratory subgroup analysis, time bias, and single-centered studies. Several on-going studies evaluating venetoclax with FLAG-IDA (NCT03214562), CPX-351 (NCT03629171), and CLIA (NCT02115295) have shown promising early results.
References
Please indicate your level of agreement with the following statements:
The content was clear and easy to understand
The content addressed the learning objectives
The content was relevant to my practice
I will change my clinical practice as a result of this content
Your opinion matters
Which AML-related topic do you currently need the most practical guidance on?

